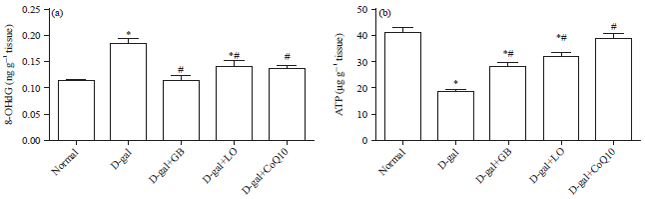
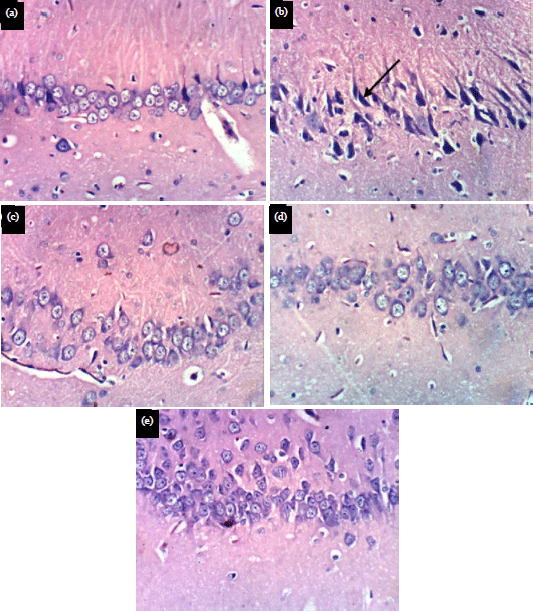
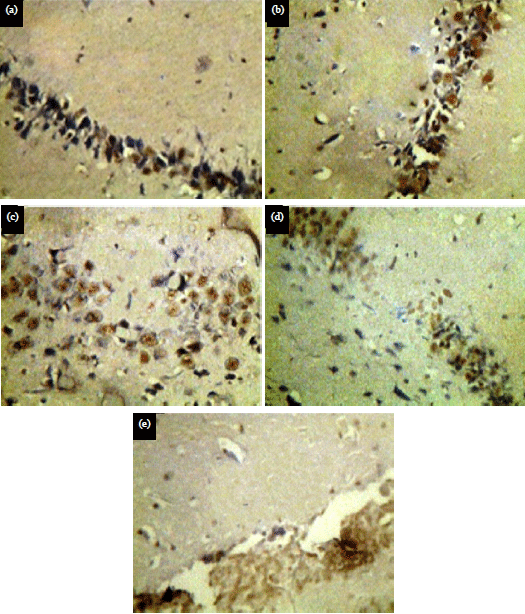
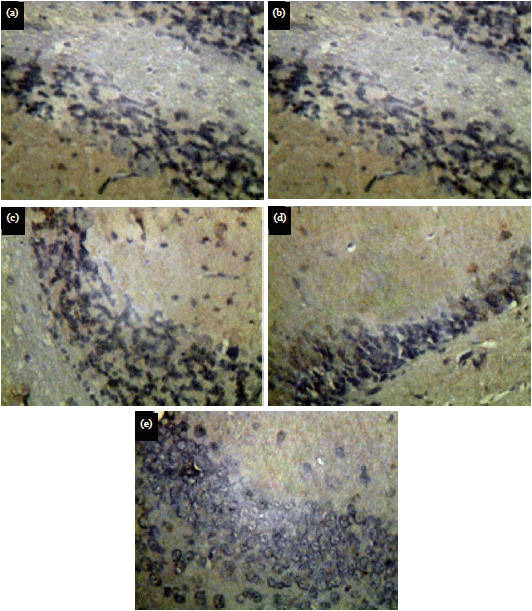
Research Article
Neuromodulatory Influences of Nutraceuticals on D-galactose Induced Hippocampal Neuronal Metabolic Dysfunction Model
Department of Pharmacology, National Organization for Drug Control and Research (NODCAR), Giza, Egypt
Omar A. Ahmed-Farid
Department of Physiology, National Organization for Drug Control and Research (NODCAR), P.O. Box 29, Giza, Egypt
LiveDNA: 20.31381
Hanan A. Rizk
Department of Pharmacology, National Organization for Drug Control and Research (NODCAR), Giza, Egypt