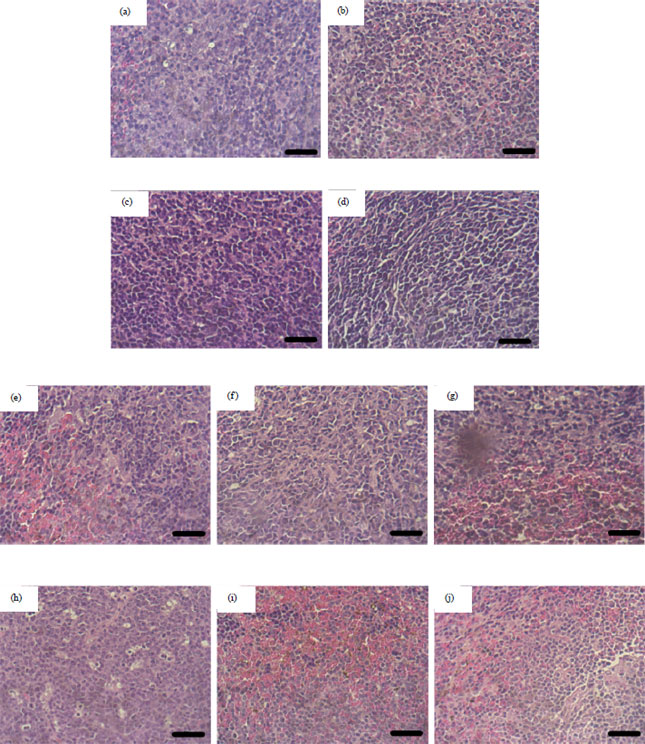
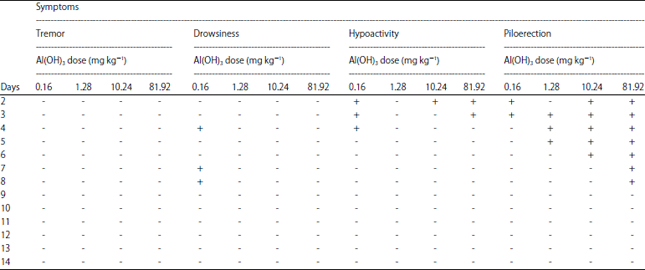
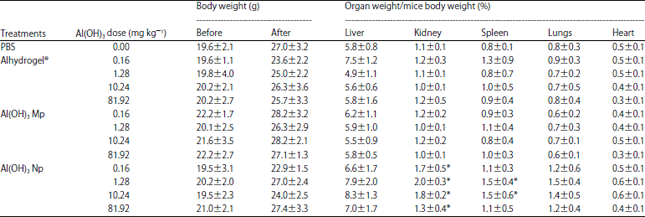
Research Article
Intraperitoneal Acute Toxicity of Aluminum Hydroxide Nanoparticle as an Adjuvant Vaccine Candidate in Mice
Centre for Pharmaceutical and Medical Technology, Agency for the Assessment and Application of Technology, Pusat Penelitian Ilmu Pengetahuan dan Teknologi, 15314 Serpong, Indonesia
LiveDNA: 62.31809
Etik Mardliyati
Centre for Pharmaceutical and Medical Technology, Agency for the Assessment and Application of Technology, Pusat Penelitian Ilmu Pengetahuan dan Teknologi, 15314 Serpong, Indonesia
Silmi Rahmani
Centre for Pharmaceutical and Medical Technology, Agency for the Assessment and Application of Technology, Pusat Penelitian Ilmu Pengetahuan dan Teknologi, 15314 Serpong, Indonesia
Sabar Pambudi
Centre for Pharmaceutical and Medical Technology, Agency for the Assessment and Application of Technology, Pusat Penelitian Ilmu Pengetahuan dan Teknologi, 15314 Serpong, Indonesia
Siti Maimunah
Centre for Pharmaceutical and Medical Technology, Agency for the Assessment and Application of Technology, Pusat Penelitian Ilmu Pengetahuan dan Teknologi, 15314 Serpong, Indonesia
Ziah Izati Azkia
Department of Pharmacy, State Islamic University Syarif Hidayatullah, 15412 Jakarta, Ciputat, Indonesia
Nurmeilis
Department of Pharmacy, State Islamic University Syarif Hidayatullah, 15412 Jakarta, Ciputat, Indonesia
Asri Sulfianti
Centre for Pharmaceutical and Medical Technology, Agency for the Assessment and Application of Technology, Pusat Penelitian Ilmu Pengetahuan dan Teknologi, 15314 Serpong, Indonesia
Julham Efendi
Centre for Pharmaceutical and Medical Technology, Agency for the Assessment and Application of Technology, Pusat Penelitian Ilmu Pengetahuan dan Teknologi, 15314 Serpong, Indonesia
Damai Ria Setyawati
Centre for Pharmaceutical and Medical Technology, Agency for the Assessment and Application of Technology, Pusat Penelitian Ilmu Pengetahuan dan Teknologi, 15314 Serpong, Indonesia
Asutiati Nurhasanah
Centre for Pharmaceutical and Medical Technology, Agency for the Assessment and Application of Technology, Pusat Penelitian Ilmu Pengetahuan dan Teknologi, 15314 Serpong, Indonesia
Irvan Faizal
Centre for Pharmaceutical and Medical Technology, Agency for the Assessment and Application of Technology, Pusat Penelitian Ilmu Pengetahuan dan Teknologi, 15314 Serpong, Indonesia