Research Article
Modulation of Visceral Nociception by Capsaicin in Mice
Department of Pharmacology, National Research Centre, Tahrir St., Dokki, Cairo, Egypt
Visceral pain is a common form of pain that is poorly understood. Nociception, the perception of noxious stimuli, is initiated by stimuli that activate the peripheral terminals of a highly specialized subset of primary afferent neurons with unmyelinated C fiber or thinly myelinated A δ fibers. These are characterized by unique sensitivity to the excitatory and sensory blocking effects of capsaicin, the pungent principle of red hot peppers (Szolcsαnyi, 1977, 1984, 1993; Holzer, 1991). Capsaicin acts on the transient receptor potential channel vanilloid receptor subunit 1 (TRPV1) which is a molecular integrator of physical and chemical stimuli in the peripheral nociceptive terminals (Caterina and Julius, 2001). Capsaicin excites and in large doses desensitizes this subset of primary afferent neurones. When stimulated, several neuropeptides such as substance P(SP), neurokinin A(NKA), neurokinin B(NKB) and calcitonin gene-related peptide (CGRP) are released from the peripheral terminals of these primary afferents. Once released these peptides mediate local tissue responses affecting motility, secretions, vascular tone, blood flow, mast cell degranulation (Szolcsányi, 1984, 1990a; Holzer, 1988, 1992; Maggi, 1995). Peptides released from polymodal nociceptive afferents are also neurotransmitters for the transmission of nociceptive information from both somatic and visceral tissues. Nociceptive messages from the inflamed peritoneum involves neurokinins (substance P, neurokinin A, neurokinin B) (Larid et al., 2000) and also CGRP (Julia and Bueno, 1997).
Capsaicin is a popular food ingredient and consumption of highly seasoned food is a habit in many different parts of the world. It therefore looked pertinent to study the effect of capsaicin in the presence of noxious peritoneal stimulus. Capsaicin was administered orally as well as intraperitoneally to mice subjected to i.p. injection of acetic acid. The latter is one of the most commonly used models of acute visceral nociception is the acetic acid induced writhing or abdominal constriction assay in which injection of dilute acetic acid into the peritoneal cavity in mice or rat triggers abdominal constrictions as a manifestation of pain (Koster et al., 1959).
Animals
Swiss male albino mice 22-25 g of body weight was used. Standard laboratory food and water were provided ad libitum. Experiments were performed between 9 am and 3 pm. Animal procedures were performed in accordance with the Ethics Committee of the National Research Centre and followed the recommendations of the National Institutes of Health Guide for Care and Use of Laboratory Animals (Publication No. 85-23, revised 1985). Equal groups of 6 mice each were used in all experiments.
Acetic Acid-Induced Writhing
Separate groups of 6 mice each were administered vehicle or capsaicin (3.3 nM-0.33 mM; 0.02 μg-2 mg kg-1, 0.5 mL i.p.). After 30 min pretreatment interval, an i.p. injection of 0.6% acetic acid was administered (Koster et al., 1959). The effect of capsaicin (0.33 mM, 0.5 mL i.p., n = 6) administered 1 h prior to acetic acid challenge was also studied. Other groups were treated with the vehicle or capsaicin (0.03 mM-0.1 mM; 0.4-1.2 mg kg-1, 1 mL i.p., n = 6/group) 1 h prior to i.p. injection of acetic acid. In a further group of mice (n = 6) capsaicin (0.1 mM) was administered twice with 30 min interval, the last dose being given 1h prior to acetic acid injection.
In addition, the effect of orally administered capsaicin (3.3, 33 μM or 0.33 mM; 20 μg kg-1, 200 μg kg-1 or 2 mg kg-1, 0.5 mL) given 90 min prior to i.p. acetic acid was studied (n = 6/group). The control group was treated with the vehicle (n = 6). Each mouse was then placed in an individual clear plastic observational chamber and the total number of writhes made by each mouse was counted for 30 min after acetic acid administration.
Further experiments were designed in an attempt to elucidate the mechanisms by which capsaicin exerts its anti-nociceptive effect. Capsaicin at concentration of 3.3 μM; 20 μg kg-1, 0.5 mL, p.o.) was selected to be used in the subsequent experiments and administered 60 min prior to nociceptive challenge with i.p. acetic acid, unless otherwise indicated.
Thus, the effect of co-administration of the α-1 adrenoreceptor antagonist prazosin (1 or 2 mg kg, s.c.) the α-2 adrenoreceptor antagonist yohimbine (5 or 10 mg kg-1, s.c.), the β-adrenoreceptor antagonist, propranolol (2 or 4 mg kg-1, s.c.), the adrenergic blocker, guanethidine (16 or 32 mg kg-1, s.c.), the muscarinic acetylcholine receptor antagonist atropine (2 or 4 mg kg-1, s.c.) were examined on antinociception caused by capsaicin. All drugs were administered 60 min prior to the abdominal constriction assay.
Furthermore, the effect of the local anaesthetic lidocaine given prior to capsaicin or vehicle was studied. Lidocaine (0.1 mL of 2% solution, p.o.) was administered immediately before or 5 min prior to 3.3 μM capsaicin (0.5 mL, p.o.) or 15 min before either 3.3 or 33 μM capsaicin (0.5 mL, p.o.). The effect of co-administerd i.p. capsaicin (3.3 μM) and i.p. lidocaine (0.1 mL of 2% solution) was also tested. Acetic acid was injected i.p. 1h after capsaicin.
In addition, the antinociceptive effect of orally or i.p. administered 3.3 μM capsaicin was studied in the presence of the mast cell stabilizer (10 or 20 mg kg-1, 0.1 mL i.p.) or cysteamine which depletes the tissues somatostatin (Szabó and Reichlin, 1981). Cysteamine (200 mg kg-1, 0.1 mL, p.o.) was co-administered with capsaicin or given as a pretreatment 60 min prior to 3.3 μM capsaicin (0.5 mL, p.o.). Acetic acid injection was carried out 1 h after capsaicin.
Drugs and Chemicals
Capsaicin, atropine sulfate, yohimbine hydrochloride, propranolol hydrochloride, guanethidine hydrochloride (Sigma, St. Louis, USA) were used. Analytical-grade glacial acetic acid (Sigma, St. Louis, USA) was diluted with pyrogen-free saline to provide a 0.6% solution for i.p. injection. All drugs were dissolved in isotonic (0.9% NaCl) saline solution immediately before use. Stock solutions of capsaicin (10 mg mL-1) contained 10% ethanol, 10% Tween 80, 80% saline solution.
Statistical Analysis
Data are expressed as mean±SE and analyzed by one way analysis of variance, followed by a Tukey's Multiple Range Test for post hoc comparison of group means. When there were only two groups a two-tailed Student's t test was used. For all tests, effects with a probability of p<.05 were considered to be significant.
Intraperitoneal Capsaicin
Capsaicin administered i.p. at increasing concentrations of 3.3 nM-0.33 mM (0.02 μg-2 mg kg-1) caused dose-dependent inhibition of abdominal constrictions induced 30 min later by i.p. acetic acid by 53.1-86.3% (Fig. 1). Marked inhibition of the nociceptive behaviour (by 86.9%) was also observed when acetic acid challenge was carried out 60 min after i.p. administration of capsaicin at 0.33 mM (Fig. 1).
Capsaicin administered i.p. at concentrations of 0.03 mM-0.1 mM (0.4-1.2 mg kg-1, i.p., 1 mL) 60 min prior to acetic acid still inhibited the nociceptive response by 94.1-97.2% when compared to the vehicle treated group (Fig. 2). Repeated capsaicin (0.1 mM) administration with 30 min interval inhibited the nociceptive response by 84.3% (Fig. 2).
Oral Capsaicin
The antinociceptive effect of capsaicin was also observed when the agent was given orally 90 min prior to the i.p. administration of acetic acid. Thus capsaicin at concentration of 3.3, 33 μM or 0.33 mM (20, 200 μg kg-1 or 2 mg kg-1, 0.5 mL, p.o.) inhibited the abdominal constrictions by 36.8-52.7% (Fig. 3).
Effect of Prazosin, Yohimbine or Guanethidine
Figure 4 shows that co-treatment with the α(1) adrenoreceptor prazosin, the α(2)-adrenoreceptor antagonist yohimbine or adrenergic neurone blocker guanethidine enhanced the antinociceptive effect of orally administered capsaicin (3.3 μM; 20 μg kg-1, 0.5 mL).
Yohimbine administered alone increased the nociceptive response, whereas prazosin and guanethidine reduced the number of abdominal constrictions induced by i.p. acetic acid (data not shown).
Effect of Propranolol or Atropine
Similarly, the non-selective beta adrenoreceptor antagonist propranolol at 2 or 4 mg kg-1, s.c., or the non-selective muscarinic receptor antagonist atropine (2 or 4 mg kg-1, s.c.) rather enhanced the effect of capsaicin in the abdominal constriction assay (Fig. 5). Propranolol itself (2 and 4 mg kg-1) inhibited the abdominal constriction response by 20 and 36.2%, respectively (54±3.9 and 44±3.1 vs 69±6.1).
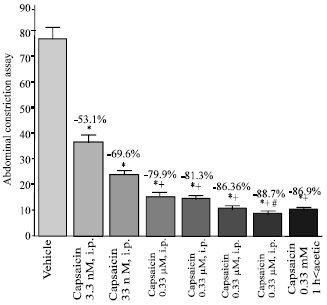 | |
| Fig. 1: | Effect of intraperitoneal capsaicin on the acetic acid-induced nociceptive responses. Mice received vehicle (control, 0.5 mL, n = 6) or capsaicin (3.3-0.33 mM, 0.5 mL (0.02 μg-2 mg kg-1) n = 6/group) 30 min prior to the test. Capsaicin (0.33 mM) was in addition administered 1 h prior to acetic acid challenge (last column). Data expressed as mean±SE and percent inhibition (%) compared to the vehicle-treated group. *p<0.05 vs. vehicle. +p<0.05 vs 3.3 or 33 nM capsaicin. #p<0.05 vs 0.33 μM capsaicin |
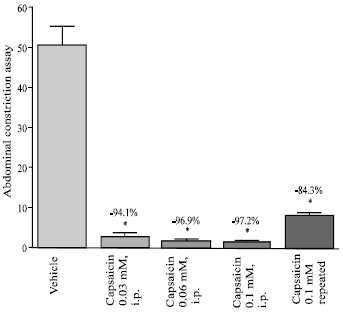 | |
| Fig. 2: | Effect of intraperitoneal capsaicin (0.03-0.1 mM; 0.4-1.2 mg kg-1, 1 mL, n = 6/ group) on abdominal constrictions caused by i.p. injection of acetic acid in mice. Capsaicin or vehicle was administered 1 h prior to acetic acid injection. Capsaicin (0.1 mM) was in addition administered twice with 30 min interval, the last dose being given 1 h prior to acetic acid challenge (last column). Data expressed as mean±SE and percent inhibition (%) compared to the vehicle-treated group. * p<0.05 (vs. vehicle) |
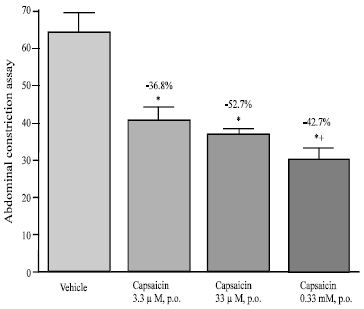 | |
| Fig. 3: | Effect of orally administered capsaicin on abdominal constrictions caused by i.p. injection of acetic acid in mice. Capsaicin (3.3, 33 μM or 0.33 mM; 20 μg kg-1, 200 μg kg-1 or 2 mg kg-1, 0.5 mL) or vehicle was administered 90 min prior to acetic acid. Data represent mean±SE and percent inhibition (%) compared to the vehicle-treated group. *p<0.05 compared to vehicle. +p<0.05 vs 3.3 μM capsaicin-treated group |
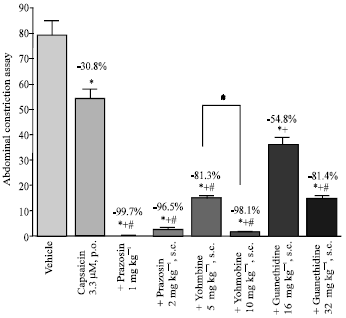 | |
| Fig. 4: | Effect of prazosin (1 or 2 mg kg-1, s.c., n = 6), yohimbine (5 or 10 mg kg-1, s.c., n = 6), guanethidine (16 or 32 mg kg-1, s.c., n = 6) on antinociception induced by orally administered capsaicin (3.3 μM; 20 μg kg-1, 0.5 mL, n = 6) in the abdominal constriction assay. Drugs were administered 60 min prior to the test. Data represent mean±SE and percent inhibition (%) compared to the vehicle-treated group. *p<0.05 compared to vehicle and between different groups as shown in the figure. + p<0.05 compared to 3.3 μM capsaicin-treated group. #p<0.05 compared to guanethidine 16 mg kg-1 + capsaicin-treated group |
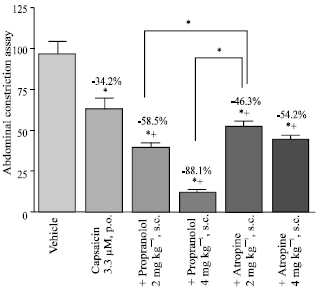 | |
| Fig. 5: | Effect of propranolol (2 or 4 mg kg-1, s.c., n = 6) or atropine (2 or 4 mg kg-1, s.c., n = 6) on antinociception induced by orally administered capsaicin (3.3 μM; 20 μg kg-1, 0.5 mL, n = 6) in the abdominal constriction assay. Drugs were administered 60 min prior to the test. Data represent mean±SE and percent inhibition (%) compared to the vehicle-treated group. *p<0.05 compared to vehicle and between different groups as shown in the figure. +p<0.05 compared to 3.3 μM capsaicin-treated group |
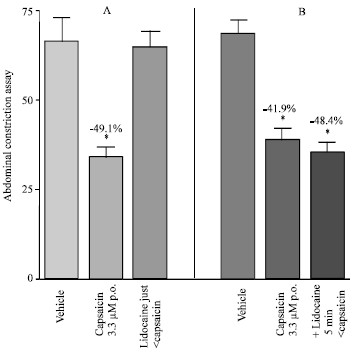 | |
| Fig. 6: | Effect of lidocaine (2%, 0.1 mL, p.o., n = 6) on antinociception induced by orally administered capsaicin (3.3 μM; 20 μg kg-1, 0.5 mL, n = 6) in the abdominal constriction assay. (A) Lidocaine was administered together with capsaicin 60 min prior to the test. (B) Lidocaine was administered 5 min prior to capsaicin and acetic acid challenge carried out 60 min later. Data represent mean±SE and percent inhibition (%) compared to the vehicle-treated group. *p<0.05 compared to vehicle |
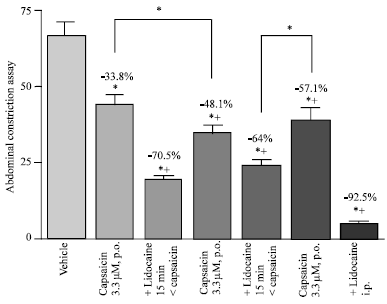 | |
| Fig. 7: | Effect of lidocaine (2%, 0.1 mL, p.o., n = 6) on antinociception induced by capsaicin in the abdominal constriction assay. Lidocaine was administered 15 min prior to oral capsaicin (3.3 or 33 μM; 20 or 200 μg kg-1, 0.5 mL, n = 6 each) and acetic acid challenge carried out 60 min later. In addition, lidocaine was administered i.p. together with i.p. capsaicin (3.3 μM; 20 μg kg-1, 0.5 mL, n = 6) (last column). Data represent mean±SE and percent inhibition (%) compared to the vehicle-treated group. *p<0.05 compared to vehicle and between different groups as shown in the figure. +p<0.05 compared to 3.3 or 33 μM capsaicin p.o.-treated group |
Atropine at 2 and 4 mg kg-1 increased visceral pain in by 19.7 and 55.8%, respectively (73±6.8 and 95±7.2 vs 61±5.3).
Effect of Lidocaine
The antinocicetive effect of orally administered capsaicin (3.3 μM ) was not observed when lidocaine was administered with capsaicin. Lidocaine given 5 min prior to capsaicin did not affect the inhibition of the abodominal constrictions by capsaicin (Fig. 6). More marked inhibition of the nociceptive response to acetic acid was observed however, when lidocaine was administered 15 min before oral introduction of capsaicin (3.3 or 33 μM) (Fig. 7). Meanwhile, lidocaine alone given orally 15 min before i.p. acetic acid injection (though not earlier) inhibited the number of abdominal constrictions by 86.4% (88±6.6 vs 12±2.3). Capsaicin (3.3 μM, i.p.) and lidocaine administered i.p. inhibited the abdominal constriction response to acetic acid by 92.5% (28.4±2.1 vs 5±0.8).
Effect of Sodium Cromoglycate
The mast cell stabilizer sodium cromoglycate administered i.p. at 10 or 20 mg kg-1, inhibited the abdominal constrictions induced by acetic acid by 68.4 and 62.9%, respectively. In mice treated with sodium cromoglycate (20 mg kg-1, i.p.), capsaicin administered i.p. (3.3 μM) (though not oral capsaicin) further reduced the nociceptive responses induced by i.p. acetic acid (Fig. 8).
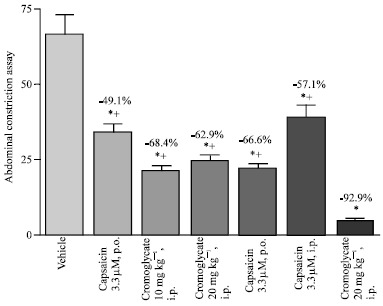 | |
| Fig. 8: | Effect of sodium cromoglycate on antinociception induced by capsaicin in the abdominal constriction assay. Sodium cromoglycate was administered i.p. together with oral capsaicin administration (3.3 μM; 20 μg kg-1, 0.5 mL, n = 6) 60 min prior to the test. In addition, sodium cromoglycate and capsaicin were i.p. administered 60 min prior to the test (last column). Data represent mean±SE and percent inhibition (%) compared to the vehicle-treated group. *p<0.05 compared to vehicle. +p<0.05 compared to cromoglycate + i.p. capsaicin 3.3 μM-treated group |
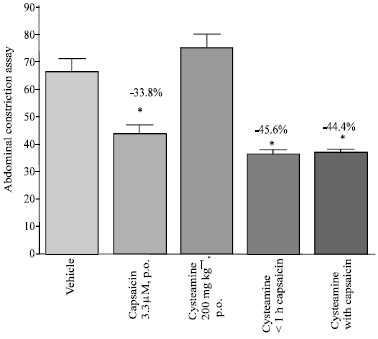 | |
| Fig. 9: | Effect of cysteamine on antinociception induced by capsaicin in the abdominal constriction assay. Cysteamine (200 mg kg-1) was orally administered 1 h before or at time of oral capsaicin administration (3.3 μM; 20 μg kg-1, 0.5 mL, n = 6). Acetic acid challenge was carried out 60 min after capsaicin administration. Data represent mean±SE and percent inhibition (%) compared to the vehicle-treated group. *p<0.05 compared to vehicle |
Effect of Cysteamine
The nociceptive behaviour following i.p. acetic acid were increased in mice treated with cysteamine at the dose of 200 mg kg-1. Meanwhile, capsaicin still inhibited the acetic acid-induced abdominal constrictions in the presence of the somatostatin depletor cysteamine (Fig. 9).
The present study indicated for the first time that oral or intraperitoneal administration of low concentrations of capsaicin led to inhibition of the nociceptive behaviour induced later by noxious peritoneal stimulus (acetic acid). In interpreting these results, it is unlikely that the observed effects are due to desensitization of sensory receptors by capsaicin. At low doses (in the μg kg-1 range) or concentrations (in the nM - μM range), capsaicin exerts a powerful excitatory effect on peripheral sensory nerve endings and induces the release of neuropeptide content from the sensory nerve terminals, this effect being confined to unmyelinated (C-fibers) and thin myleinated (Aδ fibers) afferents (Szolcsαnyi 1977, 1984; Holzer, 1988, 1992). Capsaicin introduced into the rat stomach in low concentrations of 33 μM (Szolcsαnyi and Bartho, 1980), 0.13 μM - 0.13 mM (Abdel-Salam et al., 1999), 160 μM (Holzer and Lippe, 1988; Holzer et al., 1989; Holzer et al., 1990, Szolcsányi, 1990b) effectively prevented gastric mucosal injury evoked by pylorus ligation, topical acidified aspirin, 0.6 N HCl, 96% ethanol. Conversely, aggravation of the ethanol or aspirin-induced gastric mucosal damage was observed at capsaicin concentrations of 3.3 or 6.5 mM (Szolcsányi, 1990b; Abdel-Salam et al., 1995).
The observed antinociceptive effect of capsaicin was not due to sympathetic or cholinergic reflexes since the α(1) adrenoceptor prazosin, the α(2)-adrenoreceptor antagonist yohimbine or adrenergic neuron blocker guanethidine, the non-selective beta adrenoreceptor antagonist propranolol or the non-selective muscarinic receptor antagonist atropine did not inhibit the response which, in fact, became more pronounced. This might have been due to the prevention of compensatory reflexes.
Acute, inflammatory and neuropathic pain can all be attenuated or abolished by local treatment with sodium channel blockers such as lidocaine. The peripheral input that drives pain perception thus depends on the presence of functional voltage-gated sodium channels (Wood et al., 2004). In the present study, intraperitoneal lidocaine itself inhibited the nociceptive behaviours following acetic acid injection into the abdominal cavity in mice. In decerebrate, cervical spinal cord-transected rats, intravenous lidocaine inhibited visceromotor and cardiovascular reflexes and the evoked and spontaneous activity of neurons excited by colorectal distension. Intravenous lidocaine reduces the pain of postherpetic neuralgia (Rowbotham et al., 1991), whereas intraperitoneal administration of lidocaine has been used to decrease intraoperative pain in patients subjected to tubal ligation (Visalyaputra et al., 1999). The antinociceptive action of orally administered capsaicin on the acetic acid induced abdominal constrictions was not observed when the local anesthetic lidocaine was administered just prior to capsaicin. Lidocaine given 15 min prior to capsaicin, however, enhanced the inhibition of the abdominal constrictions by capsaicin. Local (but not i.v. general) anesthetics may have inhibitory actions on TRPV1 (Hirota et al., 2003). It is worth mentioning that, the capsaicin-evoked release of somatostatin, CGRP and substance P is not inhibited by lidocaine or tetrodotoxin, indicarting the existence of a sensory neuropeptide releasing process without axon reflexes (Szolcsanyi et al., 1998).
Nociceptive messages from the inflamed peritoneum involves neurokinins (substance P, neurokinin A, neurokinin B) and also CGRP. Mice deficient in tachykinin NK1 receptor gene showed profound deficits in spontaneous behavioural reactions to an acute visceral chemical irriration (intracolonic capsaicin) (Laird et al., 2000). In rats, systemic capsaicin pretreatment or intravenous administration of CGRP antagonist (hCGRP-(8-37), completely reversed the sensitizing effects of intracolonic acetic acid on abdominal contractions in response to colorectal balloon distension. Intravenous administration of CGRP increased the number of abdominal contractions in response to colorectal distension (Plourde et al., 1997). In inflammation, neurokinin receptors (NK1 and NK2) mediate the gastric emptying inhibition and visceral pain, respectively. These responses involve a release of CGRP and hCGRP-(8-37) reduced the delay in gastric emptying and abdominal contractions induced by i.p. administration of acetic acid. Furthermore, CGRP mimicked the effects of acetic acid (Julia and Bueno, 1997). The acute intraperitoneal injection of capsaicin (0.1 and 1 μ mol kg-1) decreased gastric motility by about 50-60%, the response lasting for 15-30 min (Zittel et al., 2001).
The oesophagus and stomach are supplied by primary afferent neurones located in the nodose ganglia. The stomach is also supplied by capsaicin sensitive spinal afferent fibers containing tachykinins and CGRP (Costa et al., 2004). Nociceptive information from the stomach are also conveyed to the central nervous system by capsaicin-sensitive fibers. Capsaicin stimulates, via TRPV1, extrinsic afferents of the gut and the administration of capsaicin into the lumen of the alimentary canal evokes pain in man and mice (Holzer, 2004). Noxious gastric distention induced the expression in the nucleus of the solitary tract of c-fos, a marker for activity following noxious somatic or visceral stimulation (Traub et al., 1996). Increased c-fos transcription in the nucleus of the solitary tract and the spinal cord also followed intragastric capsaicin (3.2 mM; 2 mL) administration. The nociceptive information being processed both by gastric vagal and intestinal spinal afferents (Holzer et al., 2005).
Intragastric HCl (0.5 M)-evoked c-fos induction in many subcortical nuclei and this was depressed by at least 80% five days after bilateral subdiaphragmatic vagotomy, thereby suggesting that vagal afferent input from the acid-threatened gastric mucosa leads to the activation of subcortical brain nuclei that are involved in emotional, behavioral, neuroendocrine, autonomic and antinociceptive reactions to a noxious stimulus (Michl et al., 2001). Noxious gastric stimulation with acid (0.5 M HCl) induces release of glutamate, SP and CGRP from capsaicin-sensitive sensory afferents in the dorsal horn of the spinal cord where they may play an important role in gastric nociception and hyperalgesia (Schicho et al., 2005).
In light of the above, it is suggested that the capsaicin-induced reduction of the nociceptive behaviour following intraperitoneal administration of acetic acid is due to stimulation of capsaicin sensitive sensory afferent and transmission of nociceptive information centrally leads to the activation of descending antinociceptive pathway to a noxious stimulus.
The author acknowledges Wael Zedan and Mohamed Abd-Elaziz for their technical assistance in the experiments.