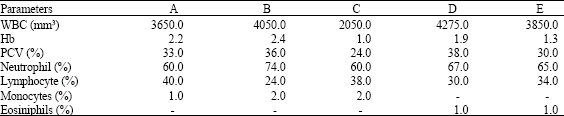
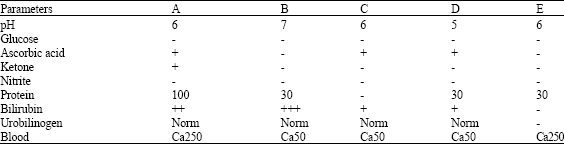
Research Article
Antiinflammatory Activity of Ethanolic Leaf Extract from Carica papaya in Rats Orogastrically Dosed with Salmonella typhi and Staphylococcus aureus
Department of Microbiology, School of Sciences, Federal University of Technology, P.M.B. 704, Akure, Nigeria
I. B. Osho
Department of Animal Productions and Health, School of Agriculture and Agricultural Technology, Federal University of Technology, P.M.B. 704, Akure, Nigeria