Research Article
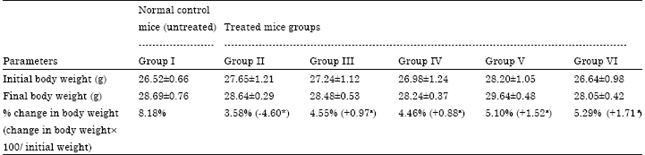
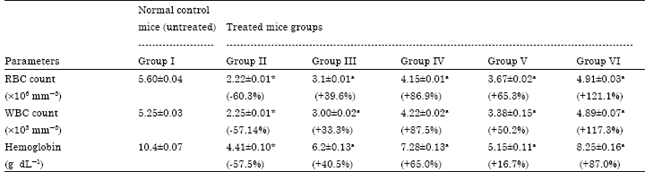
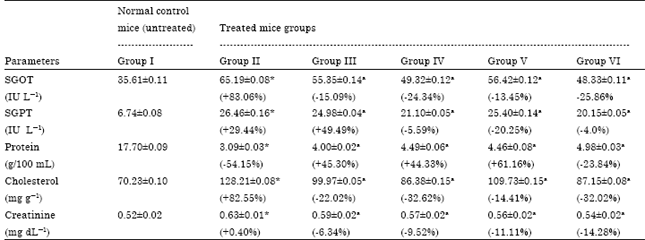
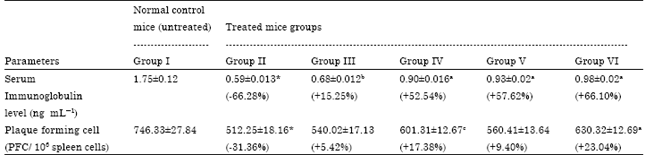
Ameliorating Effect of Coriandrum sativum Extracts on Hematological and Immunological Variables in an Animal Model of Lead Intoxication
Department of Bioscience and Biotechnology, Banasthali University, Banasthali, M.A.I. Jaipur, India
Leena Kansal
Department of Bioscience and Biotechnology, Banasthali University, Banasthali, M.A.I. Jaipur, India
Arti Sharma
Department of Bioscience and Biotechnology, Banasthali University, Banasthali, M.A.I. Jaipur, India
Shweta Lodi
Department of Bioscience and Biotechnology, Banasthali University, Banasthali, M.A.I. Jaipur, India
S.H. Sharma
Department of Bioscience and Biotechnology, Banasthali University, Banasthali, M.A.I. Jaipur, India