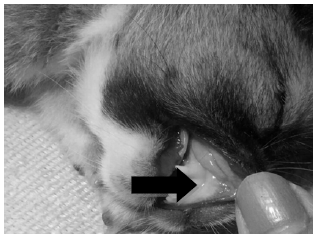
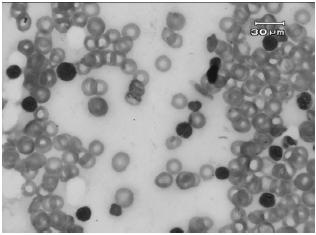
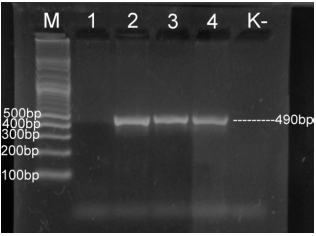
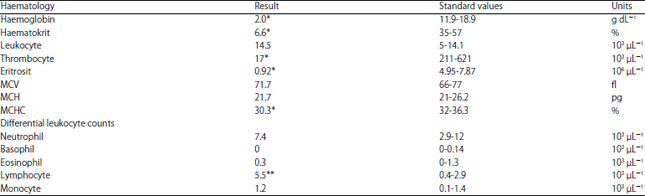
Case Study
Babesiosis in a Local Dog in Yogyakarta, Indonesia, a Case Report
Department of Parasitology, Faculty of Veterinary Medicine, Gadjah Mada University, Jl. Fauna No. 2 Karangmalang, Yogyakarta, Indonesia
Yudhi Ratna Nugraheni
Department of Parasitology, Faculty of Veterinary Medicine, Gadjah Mada University, Jl. Fauna No. 2 Karangmalang, Yogyakarta, Indonesia
LiveDNA: 62.20110
Eryl Sri Rohayati
Department of Parasitology, Faculty of Veterinary Medicine, Gadjah Mada University, Jl. Fauna No. 2 Karangmalang, Yogyakarta, Indonesia
Joko Prastowo
Department of Parasitology, Faculty of Veterinary Medicine, Gadjah Mada University, Jl. Fauna No. 2 Karangmalang, Yogyakarta, Indonesia