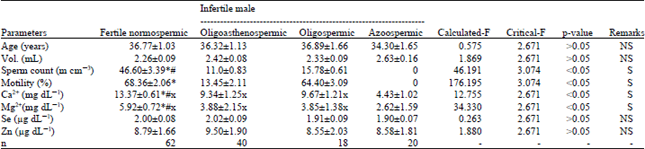
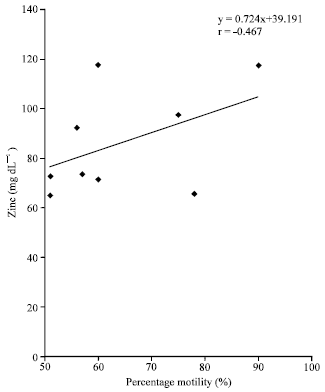
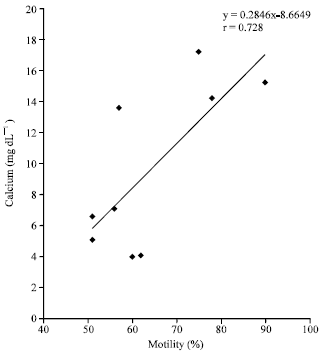
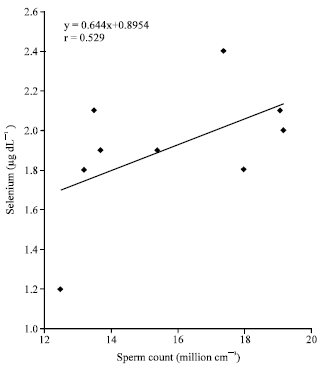
Research Article
Seminal Plasma Selenium, Calcium, Magnesium and Zinc Levels in Infertile Men
Department of Medical Laboratory Science, Chemical Pathology Unit, University of Calabar, Calabar, Nigeria
O.E. Essien
Department of Medicine, University of Calabar Teaching Hospital, Calabar, Nigeria
A.E. Udoh
Department of Medical Laboratory Science, Chemical Pathology Unit, University of Calabar, Calabar, Nigeria
I.U. Imo
Department of Medical Laboratory Science, Chemical Pathology Unit, University of Calabar, Calabar, Nigeria
I.O. Effiong
Department of Medical Laboratory Science, Chemical Pathology Unit, University of Calabar, Calabar, Nigeria