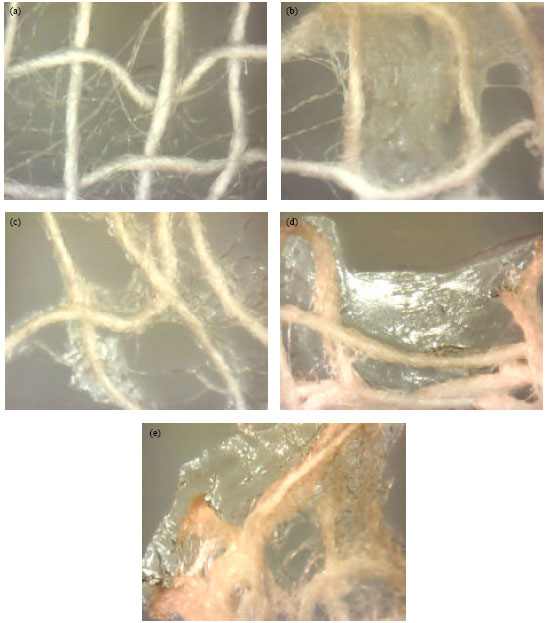
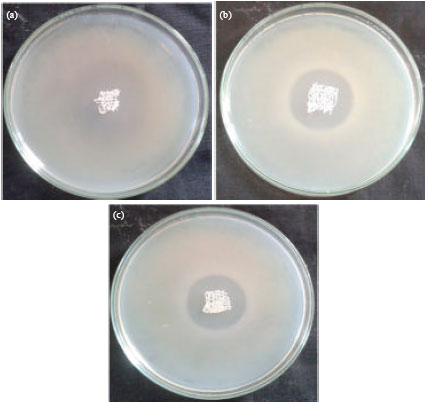
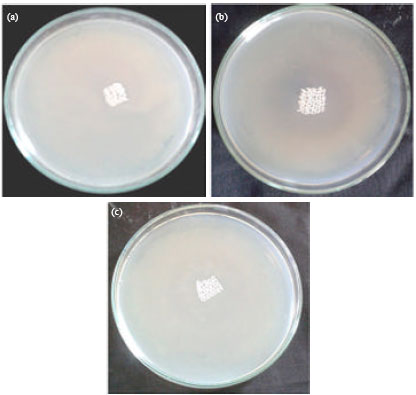
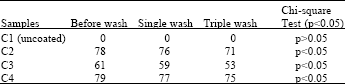Research Article
Biopolymer and Bletilla striata Herbal Extract Coated Cotton Gauze Preparation for Wound Healing
Department of Microbiology and Bioinformatics, CMS College of Science and Commerce, Coimbatore-642 006, Tamil Nadu, India
V. Vanitha Malathy
Department of Zoology, Kunthavai Naachiyar Government Arts College for Women, Tanjore, Tamil Nadu, India
B. Elayarajah
Department of Microbiology and Bioinformatics, CMS College of Science and Commerce, Coimbatore-642 006, Tamil Nadu, India
Mohan
Department of Science and Humanities, Sri Shanmuga College of Engineering and Technology, Sangagiri, Erode, Tamil Nadu, India
R. Rajendran
Department of Microbiology, PSG College of Arts and Science, Coimbatore, Tamil Nadu, India
Ram Rammohan
Department of Microbiology and Bioinformatics, CMS College of Science and Commerce, Coimbatore-642 006, Tamil Nadu, India