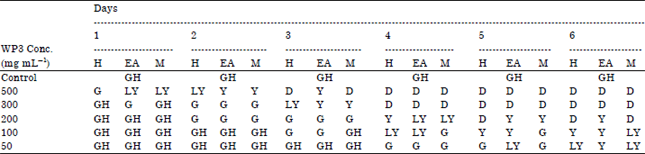
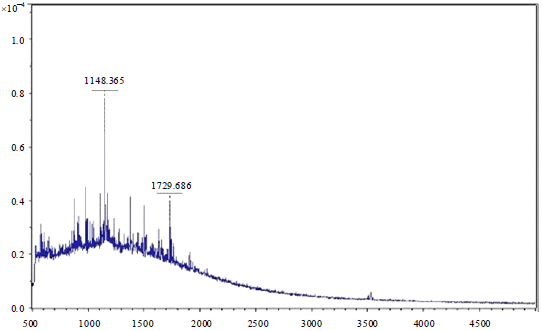
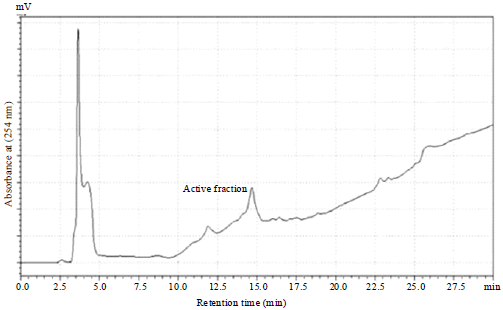
Research Article
Herbicidal Activity of an Epibiotic Bacillus Strain WP3 from Sea Fan Coral
Department of Biomedical Engineering, Vel Tech Multitech, Chennai, 600 062, Tamilnadu, India
A. Alex John
St. Peter�s University, Chennai, 600 054, Tamilnadu, India