Research Article
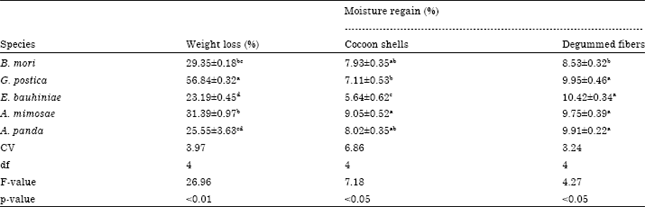
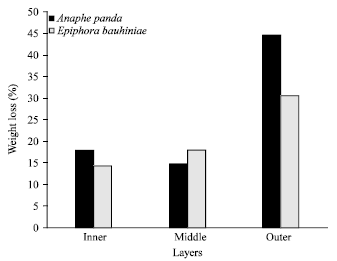
Study on Weight Loss and Moisture Regain of Silk Cocoon Shells and Degummed Fibers from African Wild Silkmoths
International Centre for Insect Physiology and Ecology, P.O. Box-30772-00100, Nairobi, Kenya
S. K. Raina
International Centre for Insect Physiology and Ecology, P.O. Box-30772-00100, Nairobi, Kenya
F. Vollrath
Department of Zoology, Oxford University, South Parks Road, Oxford OX1 3PS, United Kingdom
J. M. Kabaru
School of Biological Sciences, University of Nairobi, P.O. Box 30197-00100, Kenya
J. Onyari
School of Biological Sciences, University of Nairobi, P.O. Box 30197-00100, Kenya
E. K. Nguku
International Centre for Insect Physiology and Ecology, P.O. Box-30772-00100, Nairobi, Kenya