Research Article
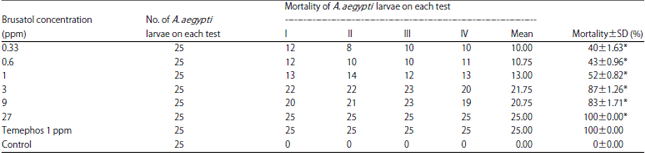
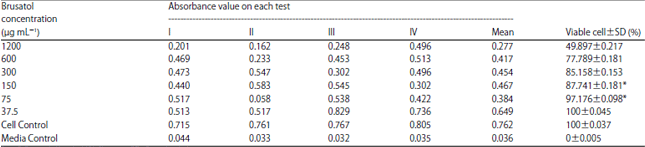
Effect of Brusatol Biolarvicide Administration on Behavioral Response of Aedes aegypti and its Toxicity on Vero Cells
Department of Epidemiology and Tropical Disease, Faculty of Public Health, University of Diponegoro, Semarang, Indonesia
LiveDNA: 62.16151
Nurjazuli
Department of Environmental Health, Faculty of Public Health, University of Diponegoro, Semarang, Indonesia