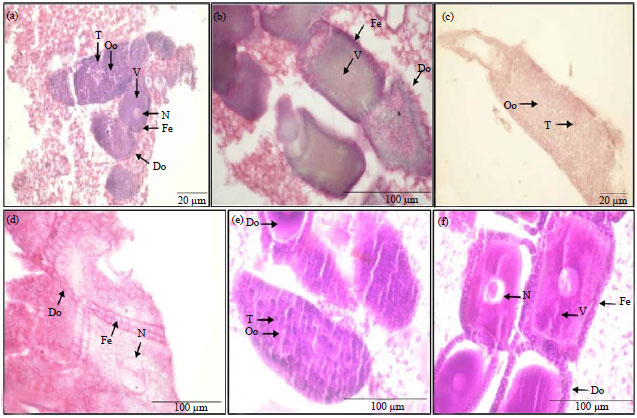
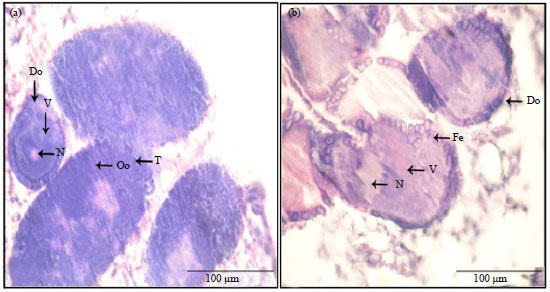
Research Article
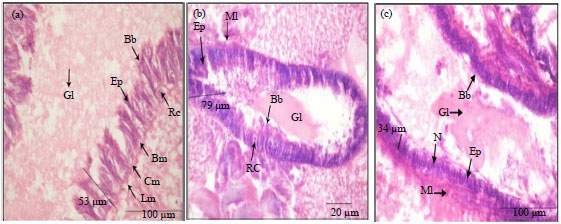
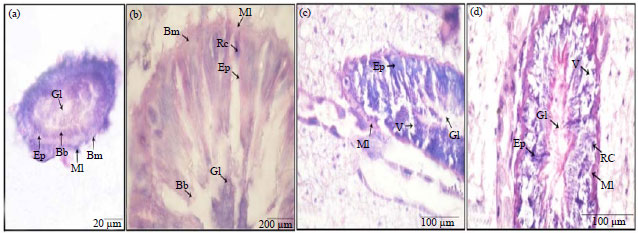
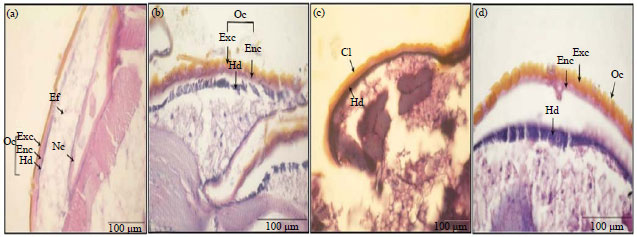
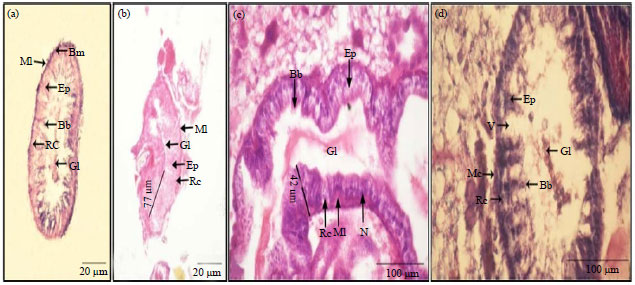
Histological Effects of Essential Oils, Their Monoterpenoids and Insect Growth Regulators on Midgut, Integument of Larvae and Ovaries of Khapra Beetle, Trogoderma granarium Everts
Department of Zoology, Faculty of Science, Alexandria University, 21511 Alexandria, Egypt
Mohamed H. Swidan
Department of Biology and Geology, Faculty of Education, Alexandria University, 21526 Alexandria, Egypt
Dalia A. Kheirallah
Department of Zoology, Faculty of Science, Alexandria University, 21511 Alexandria, Egypt
Fatma E. Nour
Department of Biology and Geology, Faculty of Education, Alexandria University, 21526 Alexandria, Egypt