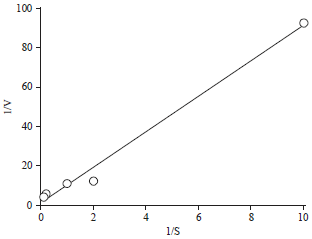
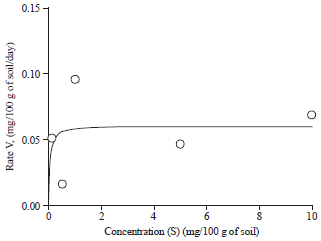
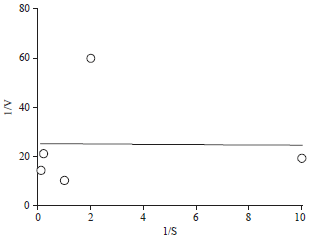
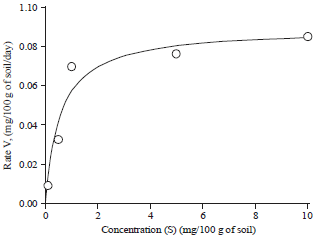
Research Article
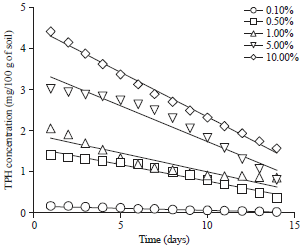
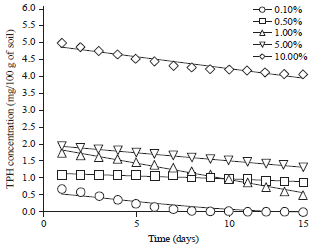
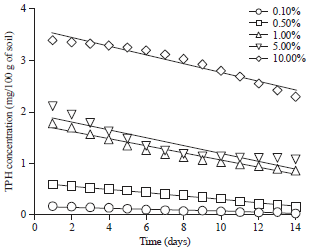
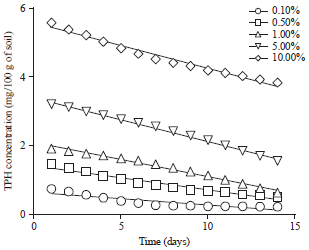
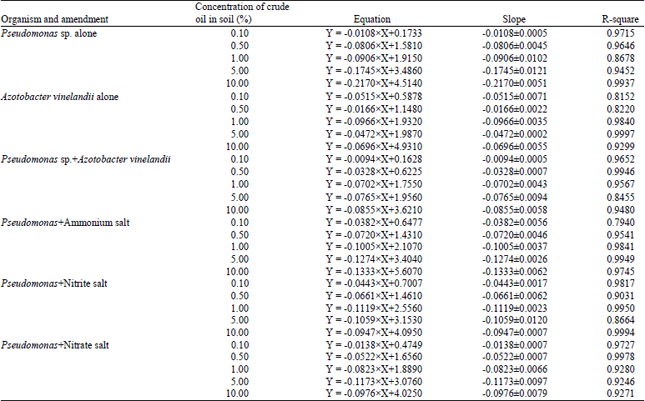
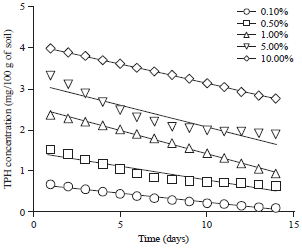
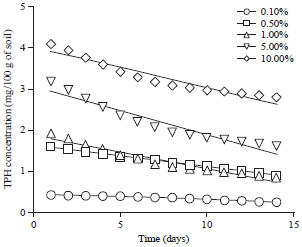
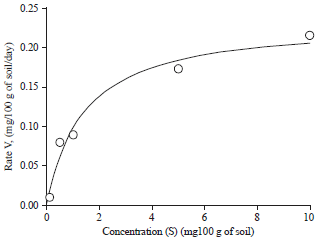
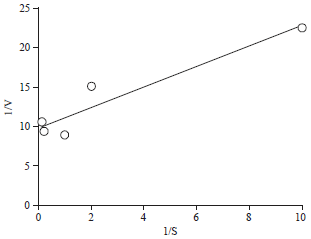
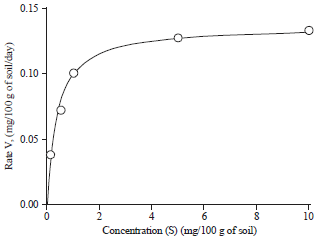
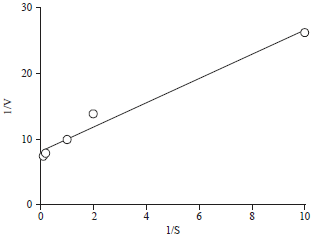
Effect of Different Organic and Inorganic Nitrogen Sources on the Kinetics of the Breakdown of Crude Oil Using Pseudomonas
Department of Chemistry, Biochemistry and Molecular Biology, Faculty of Science and Technology, Federal University Ndufu Alike Ikwo (FUNAI), Ikwo Ebonyi State, 400002, Nigeria
N.E. Onwurah Ikechukwu
Biotechnology and Pollution Control Unit, Department of Biochemistry, Faculty of Biological Sciences, University of Nigeria, Nsukka Enugu State, Nigeria
S. Ubani Chibuike
Department of Biochemistry, Faculty of Biological Sciences, University of Nigeria, Nsukka Enugu State, Nigeria
Nweke Alexandra
Department of Biochemistry, Faculty of Biological Sciences, University of Nigeria, Nsukka Enugu State, Nigeria