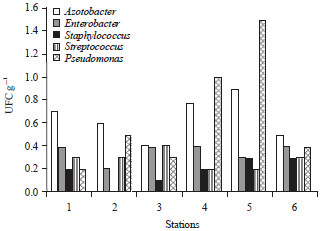
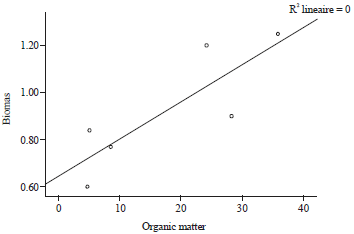
Research Article
Physicochemical and Microbiological Characterization of a Dry Soil in the Interface Steppe-Saharan Region in Southwest of Algeria
Universite de Mascara, Laboratoire, LREEG, Mascara, Algerie
Mederbal Khelladi
Faculte des Sciences de la Nature et de la Vie, Universite Ibn Khaldoun de Tiaret, Tiaret 14.000, Algerie
Regagba Mohamed
Universite de Mascara, Laboratoire, LREEG, Mascara, Algerie
Josa Ramon
Universitat Politecnica de Catalunya, UPC-ESAB, Castelldefels, Barcelone, Espagne
Daniella Meyer Allevato
University of California, Berkley, CA, USA
Latigui Amina
Faculte des Sciences de la Nature et de la Vie, Universite Ibn Khaldoun de Tiaret, Tiaret 14.000, Algerie