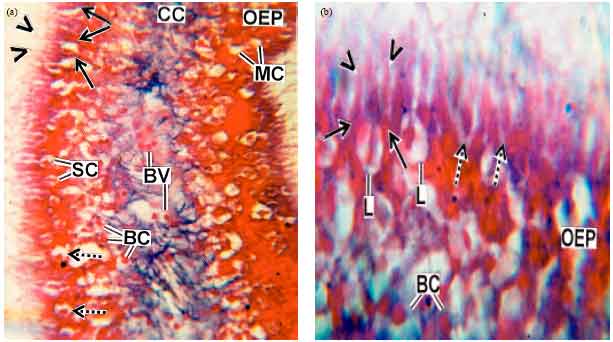
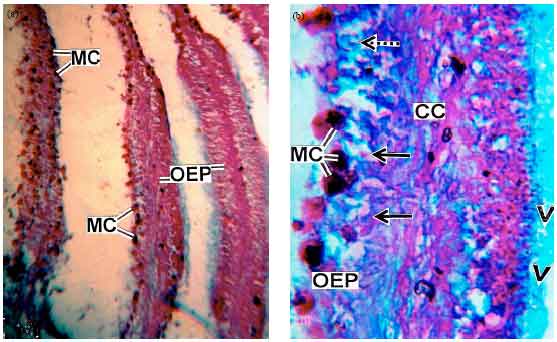
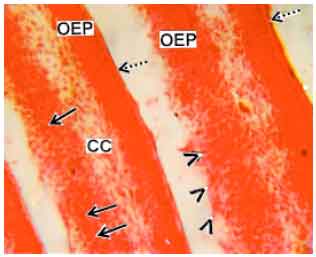
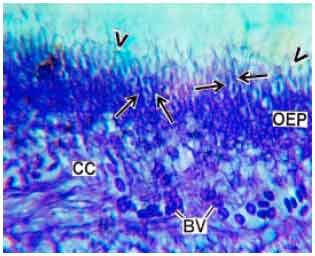
Research Article
Histological and Histochemical Studies of the Olfactory Organ in Bagrid Catfish Rita rita (Hamilton, 1822)
Fisheries Laboratory, Department of Zoology, The University of Burdwan, Burdwan 713 104, West Bengal, India
Saroj Kumar Ghosh
Fisheries Laboratory, Department of Zoology, The University of Burdwan, Burdwan 713 104, West Bengal, India