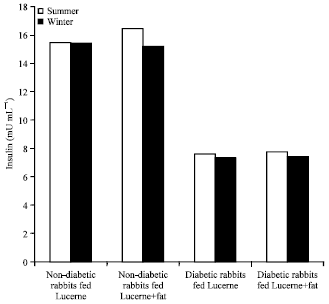
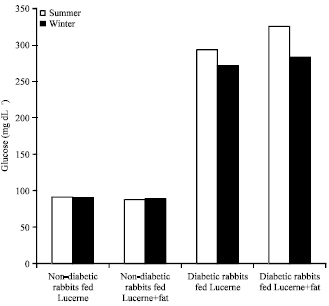
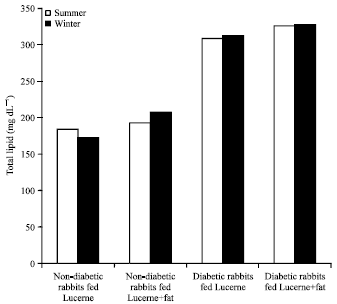
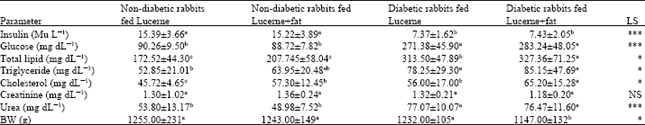
Research Article
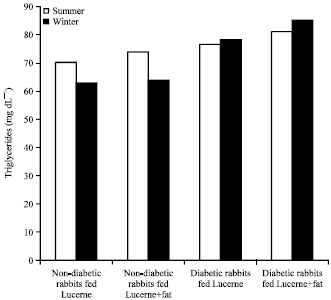
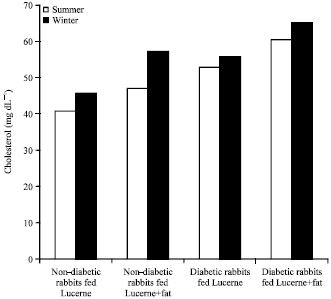
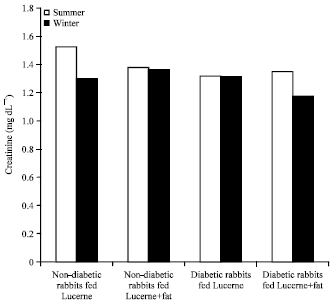
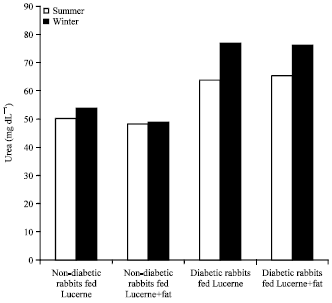
Evaluation of Effects of Milk Lipid (Ghee) Supplementation and Thermal Environment on the Metabolic Profile of Alloxan-diabetic Rabbits (Oryctolagus cuniculus)
Department of Physiology, Faculty of Veterinary Medicine, University of Khartoum, Sudan
Mariam Y. Ibrahim
Faculty of Medicine, Omdurman Islamic University, Sudan
Mohamed E. Elnageeb
Department of Physiology, Faculty of Veterinary Medicine, University of Khartoum, Sudan