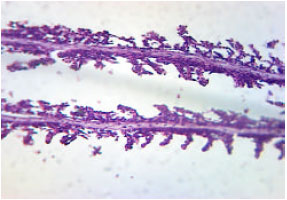
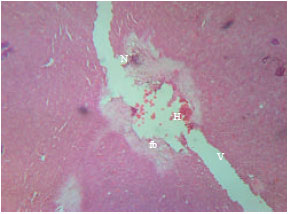
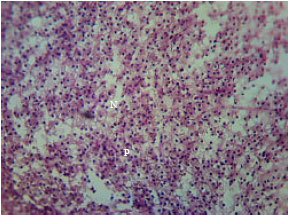
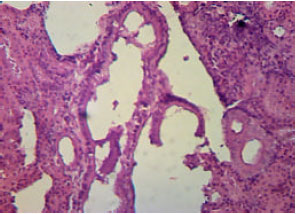
Research Article
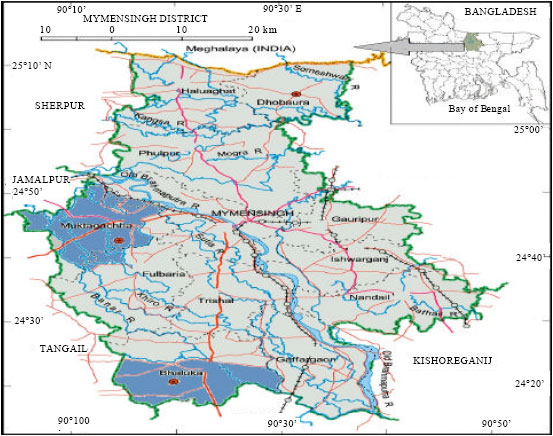
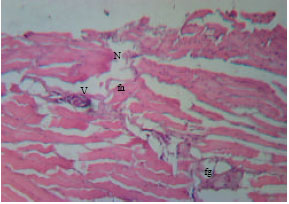
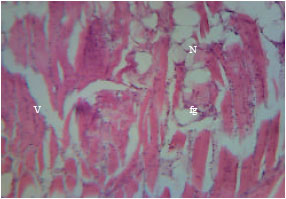
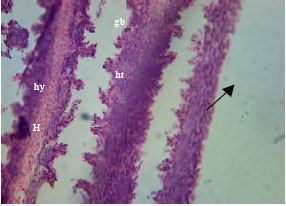
Health Condition of a Farmed Tilapia (Oreochromis niloticus) in Earthen Ponds, Northern Bangladesh
Department of Aquaculture, Bangladesh Agricultural University, 2202, Mymensingh, Bangladesh
T. Khatun
Department of Aquaculture, Bangladesh Agricultural University, 2202, Mymensingh, Bangladesh
M. Belal Hossain
Department of Biology, FOS, Universiti Brunei Darussalam, BE1410, Brunei
M. Shamsuddin
Department of Aquaculture, Bangladesh Agricultural University, 2202, Mymensingh, Bangladesh