Clinical Report
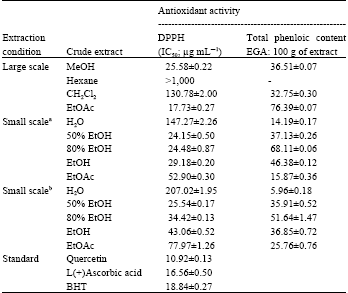
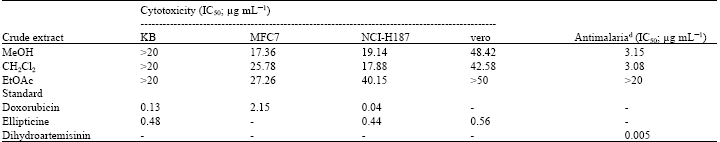
Antioxidant, Cytotoxic and Antimalarial Activities from Crude Extracts of Mushroom Phellinus linteus
Centre of Exellence for Innovation in Chemistry (PERCH-CIC), Department of Chemistry, Faculty of Science, Mahasarakham University, Maha Sarakham, 44150, Thailand
P. Seephonkai
Centre of Exellence for Innovation in Chemistry (PERCH-CIC), Department of Chemistry, Faculty of Science, Mahasarakham University, Maha Sarakham, 44150, Thailand
A. Sangdee
Department of Biology, Faculty of Science, Mahasarakham University, Maha Sarakham, 44150, Thailand
A. Puntumchai
Centre of Exellence for Innovation in Chemistry (PERCH-CIC), Department of Chemistry, Faculty of Science, Mahasarakham University, Maha Sarakham, 44150, Thailand
U. Klinhom
Department of Biology, Faculty of Science, Mahasarakham University, Maha Sarakham, 44150, Thailand