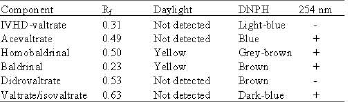
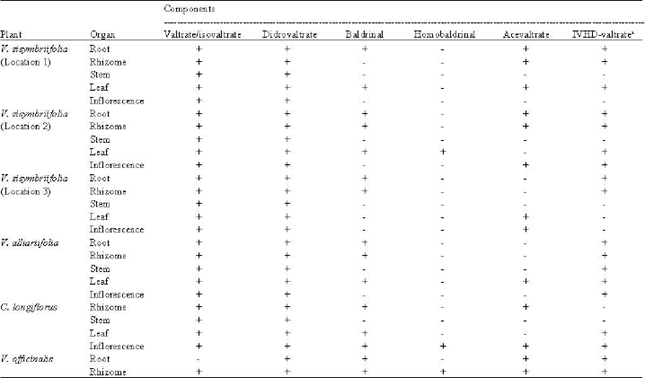
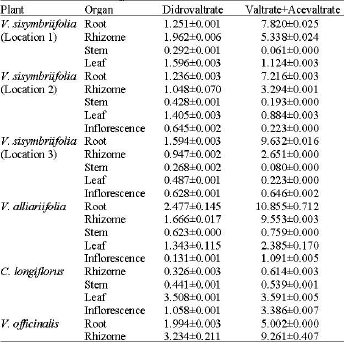
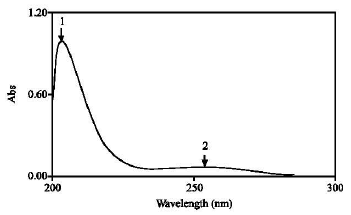
Research Article
Evaluation of Some Iranian Wild Species from Valerianaceae as Commercial Sources of Valepotriates
School of Biology, University College of Sciences, University of Tehran, Tehran, Iran
Radjabian Tayebeh
Department of Biology, Faculty of Sciences, Shahed University, Tehran, Iran
Ekhteraei Tousi Samaneh
School of Biology, University College of Sciences, University of Tehran, Tehran, Iran
Niknam Vahid
School of Biology, University College of Sciences, University of Tehran, Tehran, Iran
Mozaffarian Vali-O-Allah
Research, Education and Extension Organization, Research Institute of Forests and Rangelands, Tehran, Iran