Research Article
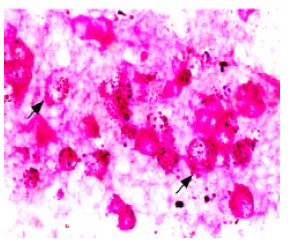
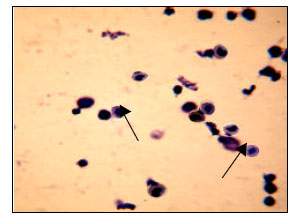
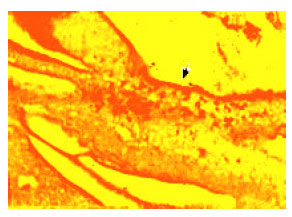
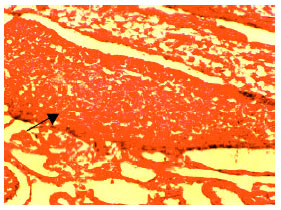
Relationship Between Spermatogonium and Testicular Development in Early Chicken Embryos
Institute of Animal Husbandry and Veterinary, Jiangsu Academy of Agricultural Science, Nanjing, 210014, China
F. Yu
Institute of Animal Husbandry and Veterinary, Jiangsu Academy of Agricultural Science, Nanjing, 210014, China
Q. Xu
Institute of Animal Husbandry and Veterinary, Jiangsu Academy of Agricultural Science, Nanjing, 210014, China
L. G. Ni
Institute of Animal Husbandry and Veterinary, Jiangsu Academy of Agricultural Science, Nanjing, 210014, China
G. H. Chen
Institute of Animal Husbandry and Veterinary, Jiangsu Academy of Agricultural Science, Nanjing, 210014, China
X.M. Cheng
Not Available
H.H. Musa
Not Available
T.Z. Liu
Not Available