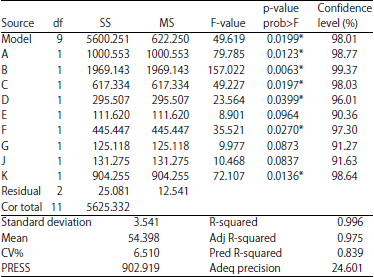
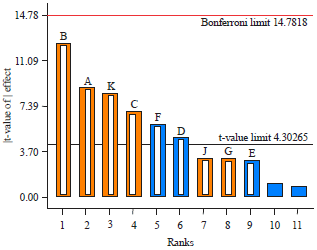
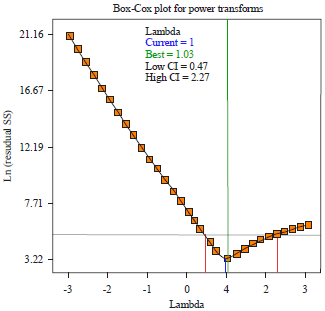
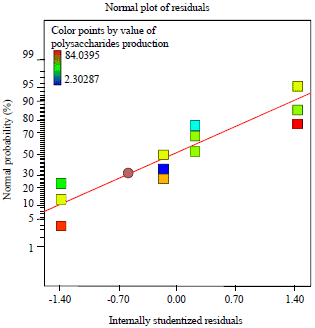
Research Article
Rapid Screening of Various Factors Influencing Polysaccharides Production by Microchlorophyte Alga, Chlorella vulgaris, by Application of Plackett-Burman Design: Characterization of Polysaccharides Using High Performance Liquid Chromatography
Department of Bioprocess Development, Genetic Engineering and Biotechnology Research Institute, City of Scientific Research and Technological Applications, Alexandria, Egypt
Mervat H. Hussein
Department of Botany, Faculty of Science, Mansoura University, Mansoura, Egypt
Sami A. Shaaban-Dessuuki
Department of Botany, Faculty of Science, Mansoura University, Mansoura, Egypt
Shimaa R. Dalal
Department of Botany, Faculty of Science, Mansoura University, Mansoura, Egypt