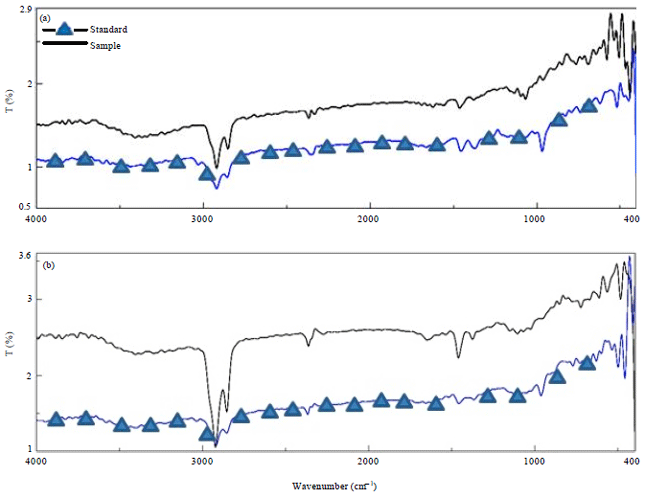
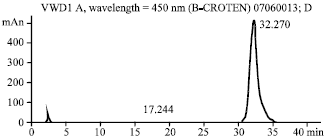
Research Article
Isolation of β-carotene from Palm (Elaeis guineensis Jacq.) Oil Using Transesterification-adsorption-desorption Method and its Characterization
Pharmacochemistry Research Group, School of Pharmacy,Faculty of Mathematics and Natural Sciences,Bandung Institute of Technology, Jalan Ganesa No.10, Bandung, 40132, Indonesia
Stefanus Andry
Physics of Electronic Materials Research Group, Faculty of Mathematics and Natural Sciences,Bandung Institute of Technology, Jalan Ganesa No.10, Bandung, 40132, Indonesi
Khairurrijal
Pharmacochemistry Research Group, School of Pharmacy,Faculty of Mathematics and Natural Sciences,Bandung Institute of Technology, Jalan Ganesa No.10, Bandung, 40132, Indonesia
Rahmana Emran Kartasasmita
Pharmacochemistry Research Group, School of Pharmacy,Faculty of Mathematics and Natural Sciences,Bandung Institute of Technology, Jalan Ganesa No.10, Bandung, 40132, Indonesia