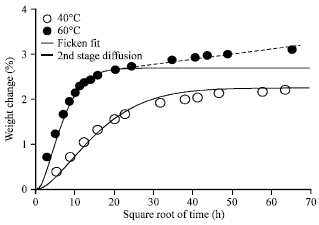
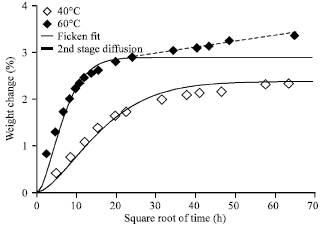
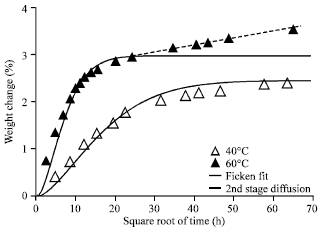
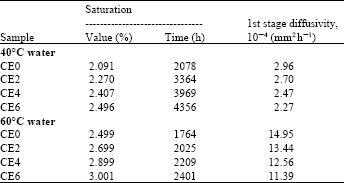
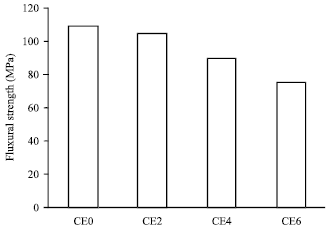
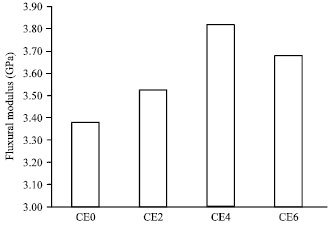
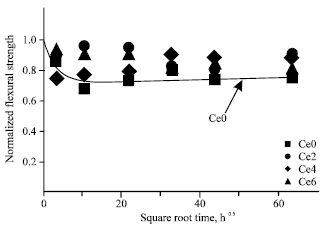
Research Article
A Study on Water Absorption and its Effects on Strength of Nano Organoclay-epoxy Composites
Faculty of Engineering Physics and Nanotechnology, University of Engineering and Technology (UET), Vietnam National University, Hanoi (VNU),144 Xuan Thuy, Cau Giay, Hanoi, Vietnam