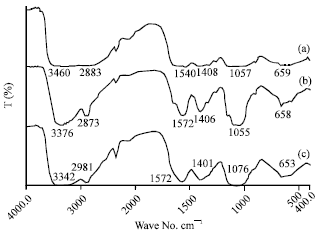
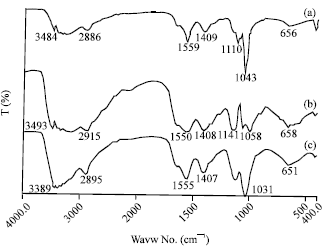
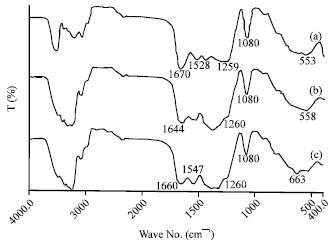
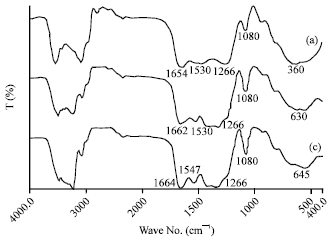
Research Article
The Effect of Temperatures and Incubation Times on Some Properties of Silk Fibroin/Chitosan Blend Films
Department of Chemistry and Center of Excellence for Innovation in Chemistry, Faculty of Science, Mahasarakham University, Maha Sarakham 44150, Thailand