Research Article
Galvanochemical Treatment Method of Copper-ammonia Bearing Rinse Wastewater
Civil Engineering Department, Mutah University 61710, P.O Box 31, Karak-Jordan
In its daily operation, the electroplating industry generates a significant amount of wastewater containing heavy metals. As a result of its higher electromigration resistance and lower resistivity, copper is used as the interconnect metal in integrated circuits replacing traditional aluminum[1]. Particularly in the production of printed circuit boards which involves, the plating and selective etching of flat circuits of copper, supported on nonconductive sheet of plastic. In this process ammonia-based etching solutions are most widely used. Use of ammonia complicates treatment of the rinse wastewater and the used electrolytes from copper[2-4]. This problem come from the chemical durability of [Cu(NH3)4]+2 complexes and the absence of an effective and reliable methods of their decomposition. Numerous methods of wastewater treatment, for ammonia based heavy metals complexes can be applied. Among these are based on the decomposition of these complexes by heating in an alkaline environment with simultaneous aeration using compressed air in order to strip out ammonia. The formed sludge is removed by sedimentation while ammonia can be absorbed by water or a solution of sulfuric acid[5].
Chemical precipitation is the most common method used for metal removal from wastewater that containing a variety of heavy metals. The basic disadvantages of all chemical methods are: The huge reagent facilities required, increasing of dissolved solids concentration in the treated water and Formation big quantities of slime in the form of badly thickened and difficulty dehydrated sludge.
Among non-chemical wastewater treatment methods from heavy metal ions, is the electrochemical method, particularly, Electro-coagulation[6,7]. The main disadvantages of this method are: Consumption of sheet metals, Passivation and coating of electrodes surface by slime, restrictions on metal concentration in the influent wastewater and high power consumption of the process.
Recently, the galvanochemical method of wastewater treatment from heavy metals, oil products and dissolved organic materials was widely used[8,9]. The method consist of wastewater treatment in reactors, where cuttings of iron and copper were added. In the reactor a contact between the cuttings and wastewater is taken place. During contacting the metals form a short circuited galvanic pair, in which iron plays a role of anode and exposed to electrochemical dissolving without using an external source of electric current. Iron dissolves in solution lead to the formation of iron oxides and hydroxides. A high treatment efficiency of wastewater from some heavy metals is reached up to 95.5-99.9% by using this method.
The main objectives of this research were:
| • | investigate the galvanochemical treatment method of copper removal, from copper-ammonia rinse wastewater. |
| • | Determine the optimum parameters of the process, such as contact time between metal cuttings and the wastewater in the reactor (Detention time), the change in wastewater pH and the effect of copper concentration in the influent wastewater on copper removal efficiency. |
| • | Determining the physical and chemical characteristics of the formed sludge (Volume, Water content, etc) and the feasibility of its reuse in the treatment, in order to increase copper removal efficiency from copper-ammonia bearing rinse wastewater. |
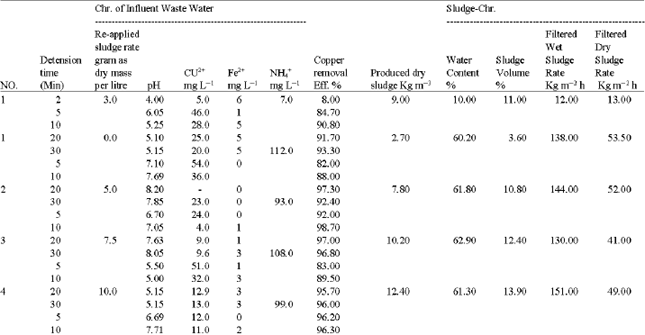
The experimental part includes two stages. In the first stage, experiments were carried out to verify the feasibility of copper-ammonia rinse wastewater treatment by galvanochemical method and to explore the optimum parameters of this method. Experiments were conducted using the experimental setup (Fig. 1). A synthetic copper-ammonia wastewater of different concentrations, 5-500 mg L-1 as Cu2+ have been prepared by dilution of concentrated copper-ammonia bearing electrolyte up to the required concentration and filled in a vessel, the water in the vessel is mixed using mechanical mixer which brought in movement by electric motor, from the vessel the solution flows by gravity to galvanic reactor volume of 0.6 L, the flow to the reactor is controlled by flow meter and the treated solution then collected in a tank. The galvanic reactor in the set up, is made of organic glass. It executed as rotating drum in order to provide a continuous contact between metal cuttings (Load) and the wastewater, it has openings for loading and unloading. The reactor supplied with six horizontal arms, to promote mixing of wastewater and the metal cuttings. It installed and supported in bearings fixed on metal frame. The rotation of the drum is carried out from electric motor which supplied with the transfer gear device with rotation velocity of 2 rpm and adjusted by the transfer device. It has a submitted voltage to the electric motor from the regulating transformer. Iron and copper cuttings in mass ratio of (Fe: Cu) = (4: 1) was used as load in the reactor. The detention time in the reactor were changed from 5-45 min in each experiment and a solution volume of 5-10 L which make the experiment duration not less than 2 h. The initial pH of the solution was adjusted by addition concentrated sulfuric acid. The synthetic wastewater was analysed before and after the treatment. The experiments were conducted with six different solution concentrations as copper (4.8, 9.5, 47, 125, 350 and 520 mg L-1 ). In each experiment prior the analysis the samples were filtered through a 45 μm glass filter to remove the sludge, The physical and chemical characteristics of the formed sludge (amount, water content and volume) were then determined. Also the filtered wet and dry sludge rates were estimated. Analysis carried out using standard methods[10,11]. Determination the produced sludge characteristics and the effect of its reuse on copper removal efficiency was obtained.
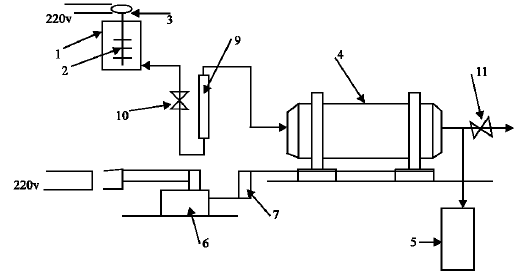 | |
| Fig. 1: | Experimental setup for the Galvano-chemical wastewater treatment 1-Vessel of influent wastewater 2-Mixer 3-Electric motor 4-Galvanic reactor 5-Tank of treated water 6-electric motor 7-Transfer gear 8-Transformer 9-flow meter 10-regulating valve 11-Sampling valve |
Experiments of the second stage were carried out on the same experimental setup Fig. 1. A copper-ammonia solution concentration of 300 mg L-13 as Cu+2, 0.58 mg L-1 Fe+2, 162 mg L-1 NH4+ and pH of 11, were used as influent. The detention time was changed from 5-30 minuets while the sludge added to the influent in eight different dosages as dry mass (0, 5, 7.5, 10, 25, 50, 75 and 100 mg L-1 ).
The experimental results of the first stage Table 1 and Fig. 2, proved the feasibility of galvanochemical method for wastewater treatment from copper. A high copper removal efficiency up to 99.3% were achieved regardless to copper concentration in the influent. The galvanochemical wastewater treatment from copper depends on the simultaneous effect of the following physical and chemical mechanisms:
| • | Dissolving of iron cuttings and formation of insoluble iron oxides and hydroxides. |
| • | Formation of insoluble copper ferrite CuFe2O3 as result of the dissolved iron reaction with copper in the solution. |
| • | Cathodic precipitation and reduction of copper cations. |
| • | Coagulation of coarsely dispersed copper impurities by iron hydroxide. |
The products of the above mentioned mechanisms were removed as a sludge by sedimentation or filtration. Despite of the change in the influent copper concentrations, which changed from 5 to 500 mg L-1 as Cu+2, the optimum detention time in the galvanic reactor should be not less than 20-30 min. The copper removal efficiency reaches 95-99%. Proceed from reference[12] and the carried out preliminary experiments the optimum pH value for the destruction of Cu-NH3 complexes is the pH in the vicinity of 6. Therefor in all experiments the pH of influent solutions was adjusted to be close to 6. As a result of pH decreasing of a solution which has concentration of 100 mg L-1 as Cu+2, was the formation of copper hydroxide flocs and the higher the concentration of copper in solution the more flocculent particles formation. The pH of treated water remains close to neutral however, using high copper concentration influent (more than 300 mg L-1) a reduction in the pH of treated water to 5-5.5 were observed. This occurs due to the hydrolysis of copper cations, which lead to copper hydroxide formation. This also could be observed by decreasing the pH of the influent.
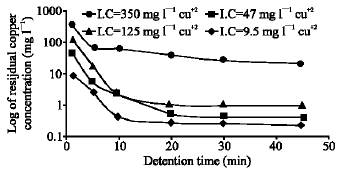 | |
| Fig. 2: | Effect of detention time on the treatment efficiency |
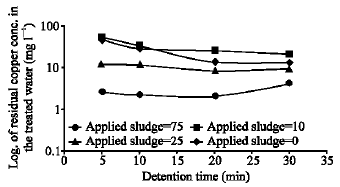 | |
| Fig. 3: | Effect of detention time on the treatment efficiency using sludge recycling as gr/L of dry mass |
It should be emphasized that this process goes more rapidly by increasing the copper-ammonia concentration in the influent solution. The formed copper hydroxide is removed with the sludge. The residual copper concentration in the treated water depends on the initial copper concentration in the influent. At influent concentrations up to 10 mg L-1 as Cu+2 the residual concentration in the treated water did not exceed 0.2-0.25 mg L-1, while at concentration up to 520 mg L-1 as Cu+2 the residual concentration remains 8-10 mg L-1. the iron concentration in the treated solution did not exceed 2-3.5 mg L-1. This is because that most of the dissolved iron in the influent and from the dissolution of iron cuttings is removed with the sludge in the form of insoluble compounds that are mainly iron oxides and hydroxide. While the concentration of ammonia in the treated water changes over a wide range, it is not affected by the pH therefore the process of ammonia strip out, has a spontaneous character and needs high control.
The results of the second stage showed that the formed sludge water content changes from 58-62%, while the filtered wet and dry sludge rates were 130-145 and 40-55 kg m-2 h-1 respectively. The results presented in Table 2 and Fig. 3 show the effect of sludge recycling on the treatment efficiency of copper-ammonia wastewater concentration of 300 mg L-1 as Cu+2. Experiments with and without sludge recycling of different dosage were carried out with detention time of 10 min.
| Table 1: | Experimental results of Glvano-chemical treatment of copper-ammonia rains wastewater |
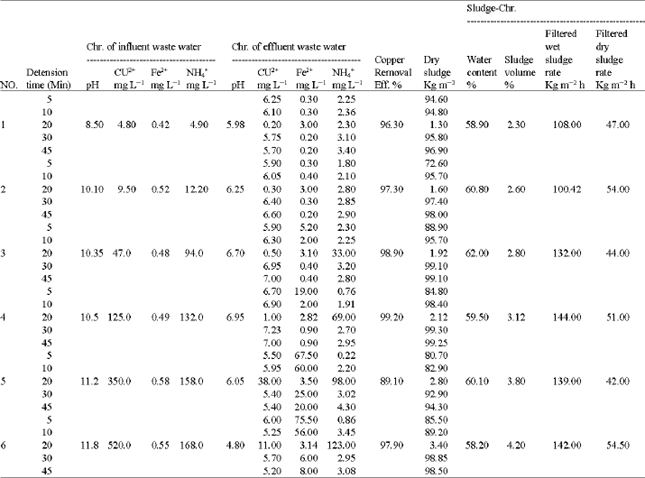 | |
| Table 2: | Experimental results for intesifcation of Glvano-chemical treatment of copper-ammonia rinse wastewater using sludge recycling |
 | |
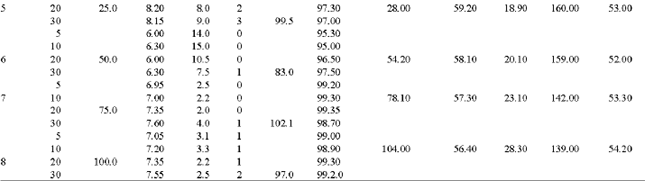 | |
The results show that the copper removal efficiency without sludge recycling reaches 90.8%, while it grows up to 99.3% by treating the same solution but with sludge recycling in amount of 75 g L-1 as dry weight. The copper removal occurs more efficient and much faster due to the increase in the contact area between the galvanic pair Fe: Cu in the reactor caused by the dispersed reduced copper in the recycled sludge and the adsorption properties of iron hydroxide in the recycled sludge. As a result of sludge recycling the required detention time reduced, from 20-30 to 5-20 min, Fig. 3. The amount of recycled sludge depends on the copper concentration in the influent the optimum sludge amount required for copper removal were estimated to be 80-250 mg of dry sludge per 1 mg L-1 of Cu+2 preferably 150-200 mg per 1 mg L-1 of Cu+2.
| • | The carried out experimental research has proved the feasibility of copper-ammonia rinse wastewater treatment by galvanochemical method, copper removal efficiency up to 99.3% were achieved using drum-type galvanic reactor. |
| • | The optimum parameters were explored: The detention time in the reactor was 20-30 min. The best influent pH where the destruction of copper-Ammonia complexes occurs more efficient were found to be close to 6. |
| • | The characteristics of the formed sludge were determined and the process of its reuse in the treatment was investigated. |
| • | The results of galvanochemical treatment of copper-ammonia bearing wastewater by sludge recycling showed that the treatment efficiency from copper increases with simultaneous reduction in the required detention time from 20-30 to 5-20 min. |