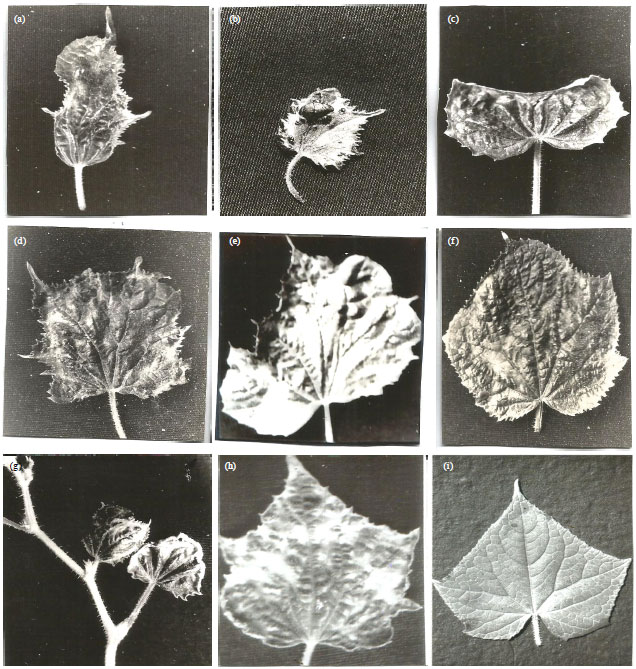
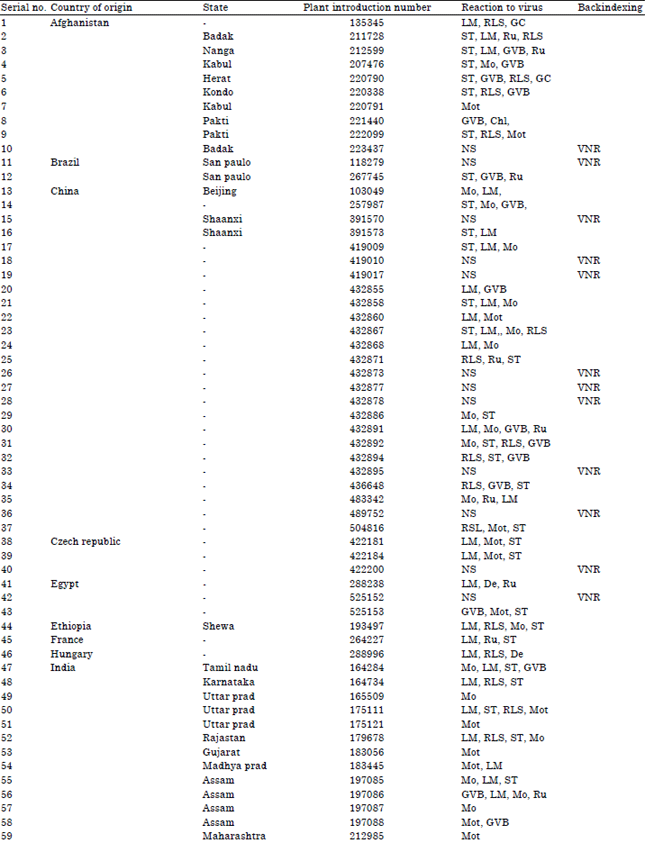
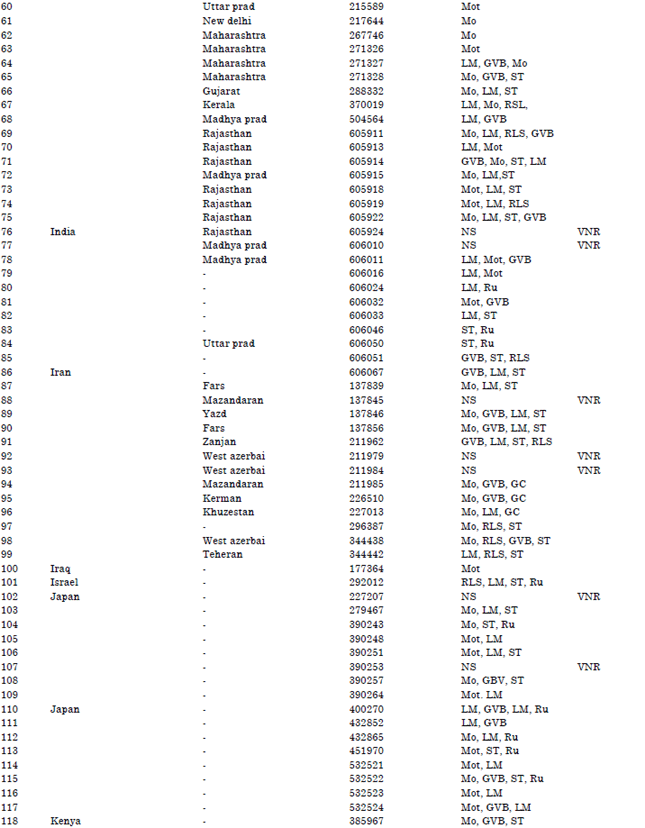
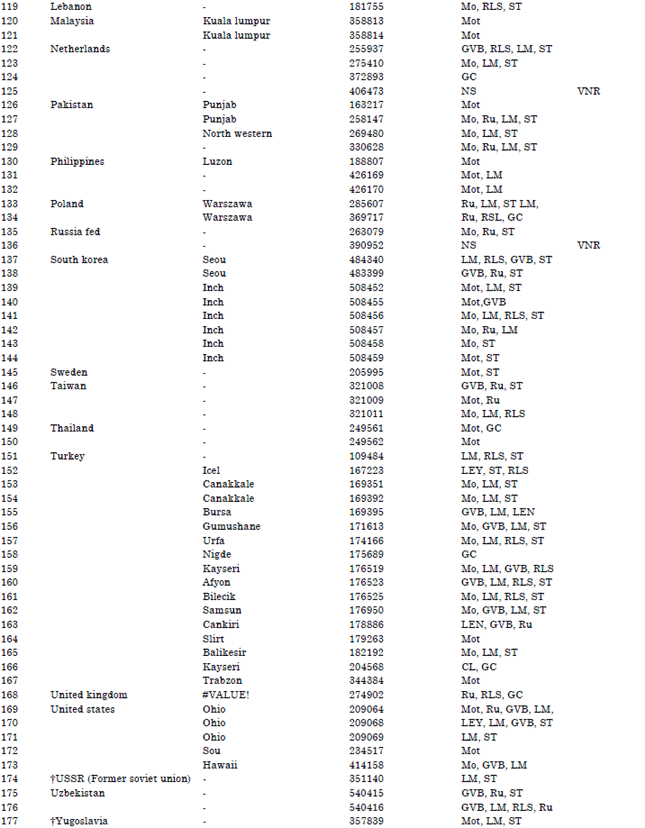
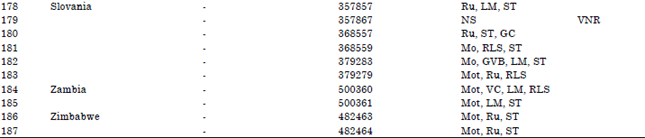
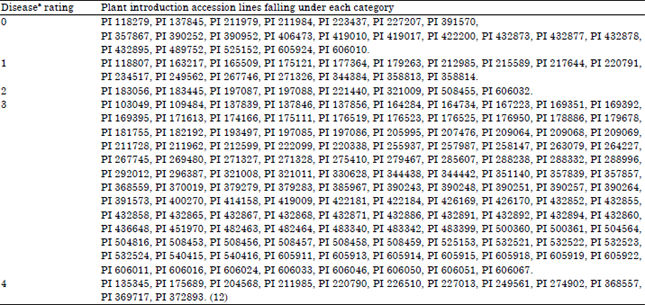
Research Article
Screening Cucumber Plant Introduction Accession Lines for Resistance against Cucumber Strain of Papaya ringspot virus (PRSV)
Department of Botany, University of Calabar, PMB 1115, Calabar, Nigeria
C.O. Nwachukwu
Department of Botany, University of Calabar, PMB 1115, Calabar, Nigeria
S. Odok
Department of Botany, University of Calabar, PMB 1115, Calabar, Nigeria