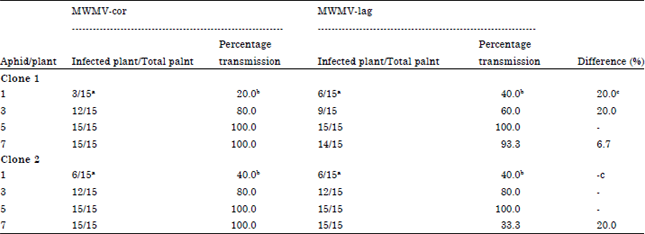
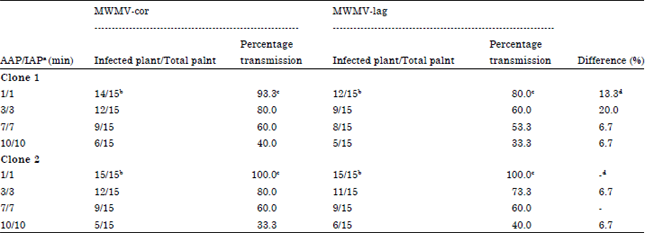
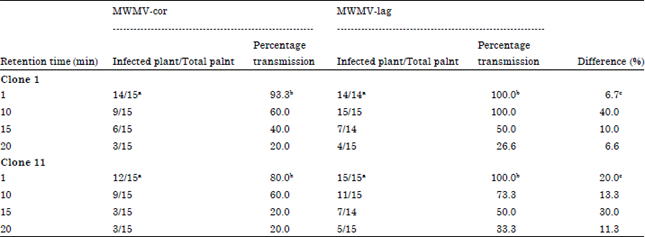
Research Article
Transmission Efficiency of Two Strains of Moroccan Watermelon Mosaic Virus by Two Clones of Aphis spiraecola (Patch)
Department of Botany, University of Calabar, PMB 1115, Calabar, Nigeria
E.E. Ekpiken
Department of Botany, University of Calabar, PMB 1115, Calabar, Nigeria