ABSTRACT
Background and Objective: The eventual switch to Light-emitting diode (LED) light fixtures as the standard in the poultry industry has resulted in the need to re-evaluate the standard management practices relating to lighting. Comparing the effect of LEDs to compact fluorescent lighting (CFL) on production and animal welfare parameters are needed. Also with the flexibility of LED lights placement of lighting can be altered to possibly maximize production and welfare by making light uniform throughout the housing system. To determine if LEDs reduce stress and fear in broilers and to see how the placement at cage level could also impact these measures. Methodology: Three experiments were conducted. The first compared rearing layers under CFL or LED lighting. The second compared using LEDs traditionally at ceiling height versus on the cage. Finally, the last experiment compared white versus red LEDs as well as utilizing the red LEDs at cage level. Results: No differences were observed in production or egg quality between CFL and LED lighting (p>0.05). Layers reared under CFL lighting exhibited greater (p<0.05) fear during tonic immobility and greater (p<0.05) stress susceptibility compared to those reared under LED lighting. In experiment 2, rearing birds with the LEDs located on the cages or in the ceiling did not appear to impact early egg production or quality. However, in experiment 3 the percentage of hens in lay was affected by not only the spectrum of LED lighting but the placement as well with red cage birds having the most birds in lay. In experiment 2, having the light on the cage did not affect fear response. While in birds appeared more stress susceptible in experiment 2 when lighting was at the cage level, this was not true in experiment 3. In experiment 3, reddish hued LED lighting reduced stress susceptibility when it was a ceiling or cage level compared to white LED lighting. Conclusion: These results indicate that during the early lay period LED lighting can reduce fear and stress responses and placement and spectrum of the lighting can also affect these responses. Finally, these results demonstrated that reddish hued LED lighting can increase the percent of hens in lay during the start of lay.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ijps.2018.92.99
URL: https://scialert.net/abstract/?doi=ijps.2018.92.99
INTRODUCTION
The poultry industry is currently undergoing a switch from traditional lighting sources like incandescent and fluorescent to light emitting diode (LED). The main driver of the switch is decreased energy usage, but LEDs can also increase bird performance and improve bird welfare via reduced fear and stress1-4. A major pitfall with the switch to a newer technology which produces a very different quality of light is that current lighting recommendations and management are based on research utilizing the older technology. As LED fixtures can be constructed in a variety of shapes and formats they will further allow for more uniform and efficient lighting in poultry houses.
Most recent research into LED lighting has focused on broiler chicken production. Studies in broilers have shown that growth and feed conversion can be affected by lighting source type. Rozenboim et al.5 and Zimmermann6 observed that there were differences between even older types of lighting technology. Mendes et al.7 found that broilers raised under LEDs performed better overall than broilers reared under CFLs. LED lighting has also been shown to improve weight gain and feed conversion in broilers over CFL lighting and incandescent lighting3,4. In laying hens, some research has not observed any effect on egg weight or hen-day egg production8 or on egg quality9 when using LED lighting versus fluorescent lighting. The spectrum of LED lights has also been observed to effect production characteristics. Min et al.10 indicated that red LED light significantly increased the thickness of eggshell compared to incandescent light. Nunes et al.11 observed increased egg weights when using red LED lights when compared to fluorescent lighting. Red LED light may also accelerate sexual development12,13 increase ovary stroma and ovarian follicle numbers14, reduce egg weight15 or increased egg weight16 and increase egg production17,18. These research findings make it necessary to re-evaluate current management of lighting if LEDs are being used.
Furthermore, different lighting types have been shown to affect fear and stress in poultry3,4. Generally, a decrease fear response has been observed in broilers chickens3,4, however, some research in laying hens have observed increase flight distances when birds are reared with LED lighting8. Fear has also been shown to be affected by different spectra of light and given that spectral output can vary drastically from light source to light source3,4. Red LED light has been observed in laying hens to increase heterophil/lymphocyte ratios and tonic immobility duration when compared to blue LED light19.
Since current management practices of lighting are based on research that used older lighting technology there is a need to reevaluate. The objectives of the 3 experiments described below was to first compare how CFL and LED lights affect layer production, egg quality, fear and stress susceptibility during the start and early lay period. The second experiments goal was to determine if placing the light source closer to the bird versus the traditional ceiling mount would affect layer production, egg quality, fear and stress susceptibility during the start and early lay period. The last experiment was designed to determine if the spectrum of light was important to the egg production and stress susceptibility during the early lay period. It is hypothesized that the use of LEDs will have beneficial effects on fear and stress responses as well as in production and that placing the fixtures at cage level will further increase the beneficial effects.
MATERIALS AND METHODS
Experiment 1: CFL versus led
Animals and husbandry: This experiment involved 2 treatments: Overdrive (LED, LsA19DIM 3000K, Overdrive, Clifton, NJ) LEDs and CFL light bulbs (CFL, Sylvania Energy Efficient CFL 2700k CF24EL, Sylvania, Wilmington, MA). Light intensity level was 20 lux at feeder level as measured with a light meter (MK350, UPRTek, Jhunan Taiwan). A comparison of spectra between these bulbs can be seen in Fig. 1. Each treatment consisted of 44 cages each containing a W-36 single comb white leghorn. Each treatment was housed within a light tight room outfitted with one of the light sources. The rooms measured 8.1×5.8 m, constructed of thick concrete walls and sealed to prevent any outside light from entering. Ventilation was provided by a single fan on the North end of the room exhausting air, which created negative pressure in the room and drew air in through cooling pads on the South wall. Each of the cages measured 304.8 mm wide×520.7 mm deep×381 mm high. The birds were managed according to the guidelines set forth in the guide for the Care and Use of Agricultural Animals in Research and Teaching20 and methods were approved by the Texas A and M institutional Animal Care and Use Committee (IACUC #2012-211 and #2012-0230). Birds were reared in from 21-31 weeks of age.
Egg production and quality: Eggs were collected daily and weighed so that average egg weight and average hen in lay could be calculated during the testing period. The eggs collected from one 24 h period during 26 and 31 weeks of age were analyzed for egg thickness using a caliper (No. 293-831, Mitutoyo, Aurora, IL) and for Haugh units using EggAnalyzer® (Orka, Bountiful, UT).
Fear tests
Novel object test: The novel object test was performed on all birds at 31 weeks of again. The birds had feed removed overnight before testing. Three birds at a time had feed returned with a small piece of pipe which was wrapped in multicolor tape placed on top of the feed. Latency for the birds to begin feeding was recorded.
Tonic immobility: Tonic immobility (TI) was conducted at 31 weeks on all bird within their housing room. Methods were modified from previous research by Archer and Mench21. A 21 cm wide by 22 cm high by 30 cm long wooden cradle with the sides sloping out at a 108 degree angle from the base was obtained, covered in a black cloth and placed on a table. Each bird was individually taken and placed on its back in the cradle. The head of the bird was covered with one hand while the breast was held with the other for approximately 15 sec to induce tonic immobility, after which time contact was removed and a timer was started. If the bird righted itself in between 15 sec, the timer was reset and the above procedure was performed again. If again the bird righted in less then 15 sec, it was recorded as a time of 0. Time of righting (or attempting to right) was recorded, with a maximum of 10 min. Longer times to righting were considered to indicate more fearfulness22.
Stress measure: Physical asymmetry of each bird was measured at 31 weeks of age, following the protocol outlined in Archer and Mench23. Using a calibrated Craftsman IP54 Digital Caliper (Sears Holdings, Hoffman Estates, IL), the middle toe length, metatarsal length and metatarsal width were measured for both the right and left legs. The composite asymmetry score was calculated by taking the sum of the absolute value of left minus right of each trait, then dividing by the total number of traits. Thus the formula for this trial would be (|L-R|MTL+|L-R|ML+|L-R|MW)/3= composite asymmetry score.
Experiment 2: Ceiling level lighting versus cage level lighting
Animals and husbandry: This experiment involved 2 treatments: Overdrive (LED ceiling, LED, LsA19DIM 3000K, Overdrive, Clifton, NJ) LEDs at 3 m from the floor and strip LED lighting placed on the top of each cage at the midpoint (LED cage, WLFA2-xW15SMD 3500K, Superbrightleds, St. Louis., MO). Light intensity level was 20 lux at feeder level as measured with a light meter (MK350, UPRTek, Jhunan Taiwan). A comparison of spectra between these bulbs can be seen in Fig. 1. Each treatment consisted of 48 cages each containing a W-36 single comb white leghorn. Each treatment was housed within a light tight room outfitted with one of the light sources.
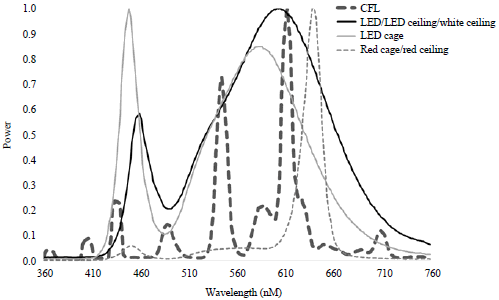 | |
| Fig. 1: | Comparison of spectrum readings used in all experiments (Experiment 1: CFL and LED, Experiment 2: LED ceiling and LED cage, Experiment 3: White ceiling, red ceiling and red cage) measured using hand held spectrometer (UPRtek, Taiwan; Model MK350) |
All animal care was conducted as in experiment 1. Birds were reared in from 21-28 weeks of age.
Egg production and quality: Eggs were collected daily and weighed so that average egg weight and average hen in lay could be calculated during the testing period. The eggs collected from one 24 h period during 22 and 28 weeks of age were analyzed for egg thickness using a caliper (Caliper: Mitutoyo, No. 293-831) and for Haugh units using the EggAnalyzer® (Orka, Bountiful, UT).
Fear tests
Novel object test: The novel object test was performed on all birds at 28 weeks in this experiment using the procedure described in experiment 1.
Tonic immobility: Tonic immobility (TI) was conducted at 28 weeks of age on all birds as described in experiment 1.
Inversion: When the birds were 28 weeks old, all birds were subjected to an inversion test, as described by Newberry and Blair24. Each bird was caught and then inverted it by holding it by its legs with one hand until the bird ceased to wing flap for 30 sec. The intensity of flapping was determined by counting the number of wing flaps and duration of flapping from video recordings (Cannon, ZR900, Melville, NY, USA; 24 frames sec–1). Greater intensity of flapping was considered to indicate more fearfulness24.
Stress measure: Physical asymmetry of each bird was measured at 28 weeks of age following the procedure described in experiment 1.
At 28 weeks of age blood samples were collected from 10 random birds per treatment via the wing vein. Between 1-2 mL of blood were collected from each bird. The blood was injected into a plasma separation gel and lithium heparin vacutainer (BD 368056, BD, Franklin Lakes, NJ), which was temporarily stored in an ice bath. Once all samples had been taken, the vacutainers were spun down in an Eppendorf AG Centrifuge 5804 (Eppendorf, Hauppauge, NY) for 15 min at 4000 rpm to separate the cells from the plasma. The plasma was poured off into 2 mL microcentrifuge tubes and stored at -19̊C until further analysis. Plasma corticosterone concentrations were measured using a commercially available ELISA kit (Enzo Life Sciences, ADI-901-097, Farmingdale, NY). The inter and intra-assay CV (%) were both under 5%.
Experiment 3: White ceiling level vs red ceiling level vs cage level red lighting
Animals and husbandry:This experiment involved 3 treatments: Overdrive (white ceiling, LSA19DIM 3000K, Roanoke, VA) LEDs or once (red ceiling, Agrishift® MLL, Once, Inc., Plymouth, MN) LEDs at 9 m above the floor or and Once 3 watt LED lighting (red cage, Agrishift® EL, Once, Inc., Plymouth, MN) placed on the top of each cage above the feed trough. Light intensity level was 20 lux at feeder level as measured with a light meter (MK350, UPRTek, Jhunan Taiwan). A comparison of spectra between these bulbs can be seen in Fig. 1. Each treatment consisted of 42 cages each containing a W-36 single comb white leghorn. Each treatment was housed within a light tight room outfitted with one of the light sources. All animal care was conducted as in experiment 1. Birds were reared in from 20-25 weeks of age.
Egg production and quality: Eggs were collected daily and weighed so that average egg weight and average hen in lay could be calculated during the testing period.
Stress measure: Physical asymmetry of each bird was measured at 25 weeks of age following the procedure described in experiment 1.
At 25 weeks of age blood samples were collected from 10 random birds per treatment as in experiment 2. Additionally, 1 mL of the blood sample was injected in to a serum separation tube (BD 367981, BD, Franklin Lakes, NJ) which was placed on its side overnight and then spun down and stored using the same procedure as in experiment 2. Plasma corticosterone and serum serotonin concentrations were measured using a commercially available ELISA kits (Enzo Life Sciences, ADI-901-097 and ADI-900-175, Farmingdale, NY). The inter and intra-assay CV (%) were both under 5% for both assays.
Statistical analysis: To investigate treatment effects on composite asymmetry, corticosterone, serotonin, novel object, inversion, tonic immobility, shell thickness, Haugh unit and average egg weight and production using the GLM procedure was used with lighting type as factor. The least significant difference test was used to test all planned comparisons. All of the assumptions were tested (Shapiro-Wilk test for normality, Levene's test for homogeneity of variance). No transformations were needed to meet assumptions. All analyses were performed using SAS 9.3 for Windows (SAS Institute Inc.). Significant differences were at p<0.05.
RESULTS
Experiment 1
Egg production and quality:The results of the egg quality and production during experiment 1 are presented in Table 1. No differences between treatments (p>0.05) in average egg weight, egg production (% hen in lay), eggshell thickness at either time point, or Haugh unit at either time point were observed.
Fear and stress responses: The results of the novel object testing, tonic immobility testing and composite asymmetry are presented in Table 1. No differences were observed between treatments (p>0.05) in latency to approach the novel object during novel object testing. Treatments did however differ in latency to right during tonic immobility with the LED birds taking less time to right than the CFL birds (p = 0.003). The LED birds also had lower composite asymmetry scores than the CFL birds (p = 0.03).
Experiment 2
Egg production and quality: The results of the egg quality and production during experiment 2 are presented in Table 2. No differences between treatments (p>0.05) in average egg weight, egg production (% hen in lay), eggshell thickness at 21 weeks of age, or Haugh unit at either time point were observed. However, the LED cage birds had thicker eggshells at 28 weeks of age when compared to the LED ceiling birds (p = 0.03).
Fear and stress responses: The results of the novel object testing, tonic immobility testing, inversion testing, composite asymmetry and plasma corticosterone concentrations are presented in Table 3. No differences were observed between treatments (p>0.05) in latency to right during tonic immobility testing or intensity of flapping during inversion testing. Treatments did however differ in latency to approach during novel object testing with the LED ceiling birds taking less time to approach than the LED cage birds (p = 0.003). While the treatments did not differ in composite asymmetry scores (p>0.05) the LED ceiling birds had lower plasma corticosterone concentrations than the LED cage birds (p = 0.001).
Experiment 3
Egg production and quality:The results of the egg quality and production during experiment 3 are presented in Table 4. No differences between treatments (p>0.05) in average egg weight, however, egg production (% hen in lay) did differ between treatments (p = 0.001).
| Table 1: | Experiment 1: Production (average egg weight, hen in lay), egg quality (shell thickness and Haugh unit at 26 and 31 weeks of age), novel object response (latency to approach), tonic immobility response (latency to right) and composite asymmetry score of layers housed under LED or CFL lighting |
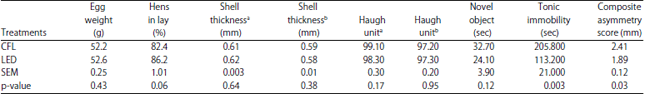 | |
aWeek 26 of age, bWeek 31 of age | |
| Table 2: | Experiment 2: production (average egg weight, hen in lay) and egg quality (shell thickness and Haugh unit at 21 and 28 weeks of age) of layers housed under LED lights located on the ceiling or on the cage |
 | |
| aWeek 21 of age, bWeek 28 of age | |
| Table 3: | Experiment 2: Novel object response (latency to approach), tonic immobility response (latency to right), inversion response (intensity of flapping), composite asymmetry score and plasma corticosterone concentrations of layers housed under LED lights located on the ceiling or on the cage |
 | |
| Table 4: | Experiment 3: Average egg weight, % hen in lay, composite asymmetry score, plasma corticosterone and serotonin concentrations of layers housed under white LED lights located on the ceiling, red LED lights located on ceiling or red LED lights on the cage |
 | |
Different letters within column indicate significant differences (p<0.05) | |
The white ceiling birds had the lowest % of hens in lay during the experiment and the red cage birds having the highest % of hens in lay; The red ceiling birds were in the middle of the other two treatments.
Stress response: The stress response data for experiment 3 is presented in Table 4. Treatments differed in all three stress measures collected (p<0.05). The white ceiling birds had the greater (p<0.05) composite asymmetry scores and plasma asymmetry scores than both the red ceiling and RED cage birds. The red ceiling birds also had higher (p<0.05) serum serotonin concentrations than both the white ceiling and red cage birds.
DISCUSSION
The 3 experiments conducted in this current study demonstrated once again that lighting can greatly impact not only production in chickens but also fear and stress responses. Experiment 1, did not demonstrate any difference between CFL and LED lighting in egg weights or egg quality during the early laying period, however, there was a significant difference in the percent of hens in lay during the experimental period. This current study did observe that there were less birds laying on average when they were housed under CFL lighting when compared to LED lighting. These results are similar to those found by Long et al.8 and Kamanli et al.9 however, contrary to what Long et al.8 over served this current study did see an increase in egg production in the LED treatment in experiment 1. Experiment 1 also observed decrease stress susceptibility in birds reared under LED lighting when compared to those reared under CFL lighting. This decreased stress susceptibility is consistent with results observed previously in broiler chickens3,4. There was also an increase in egg production in hens reared under red LED lighting when comparted to white LED lighting in experiment 3. This is consistent with other research17,18 which observed increases in production when red LED lighting used. This is likely associated with the accelerated sexual development12,13 and increased stroma and ovarian follicle numbers14. This current study did not see any effect on egg weights which may be due to the fact that this was only a brief part of the lay cycle compared to other research that looked at much longer laying periods15,16.
While pervious research has demonstrated that red LED lighting results in increased fear and stress in laying hens when compared to other spectrums of light, that was not observed in this current study. In experiment 3, rearing birds with red light located in the ceiling or on the cages resulted stress. It is possible that the differences between the results of the current study and previous research is due to the specific lighting fixtures used. In broilers, it has been observed that LEDs produced by different manufactures produce different light spectrums and that can differentially affect fear and stress responses4. It is not known what the spectrum of light was in the previous research so without that information it can only be hypothesized that this may be the reason the results differ.
Previously, no research has been conducted looking at the placement of LED lighting in laying hens. Previous research in broiler chickens has shown that placing the LED lighting close to the birds increased production and decreased fear and stress3. In experiment 2, no differences between egg production and quality was observed indicating that the location of the LED lighting was not a factor. However, fear and stress response were affected. While tonic immobility and inversion responses did not differ the novel object, test demonstrated treatment differences. The birds housed with LED lighting on the cage had longer latencies to approach the novel object. This may be because the object was better illuminated by the lighting fixture for some reason. Though the light intensity was the same in the feed troughs it is possible that because the rest of the room was dark in the LED cage treatment the object stood out more to the birds. This merits require further investigation. Furthermore, while composite asymmetry scores did not differ between treatments in experiment 2 the plasma corticosterone concentrations in the LED cage birds was significantly higher than the LED ceiling birds. These fear and stress results in experiment 2 are contrary to previous research3 in broilers which saw reduced fear and stress responses when LED lighting was positioned closer to the birds. One possible reason is the lighting was placed on top of the cages and the lighting was measured at the feeder. The light intensity was much brighter closer to the birds’ heads when they were just standing in the cage and not eating. This high intensity might have led to increased stress, high intensity light has been observed previously to increase fear and stress in poultry25. However, in experiment 3, when red LED lighting was used and was positioned over the feeders, reduced fear and stress was observed when compared to white overhead lighting. This further emphasizes that not only is positioning of the light appropriately important for maximum production and welfare that the spectrum of that light plays a role.
CONCLUSION
The experiments conducted in this study looked to determine the effects of some lighting factors during the early lay period on production, fear and stress. It is hypothesized based on work in broilers that production and fear and stress responses would be improved using LED lighting compared to CFL lighting. We also thought that positioning LED lighting close to the birds would improve these factors as well. We were not completely correct in our hypothesis. Increases in production during this early lay period using LED lights compared to CFL lighting was observed. We did however see improvements in production and fear responses when using LED lighting especially when using red LED lighting and positioning the lighting appropriately. The overall conclusion of this research is that LED lighting can be used to minimally maintain production and egg quality but it can also be used to reduce fear and stress in laying hens. Using LED lighting that contains red light is especially beneficial to production and welfare of laying hens during this early lay period. Management practices relating to lighting need to be reevaluated as LED lighting will have a large impact on how layers can be reared.
SIGNIFICANCE STATEMENT
LED lighting reduced stress and fear compared to CFL lighting. Lighting from cage level compared to the ceiling can differentially stress and fear responses depending on the location and spectrum of the light. Reddish LED lighting reduced fear and stress and using it at cage level further improved egg production.
REFERENCES
- Benson, E.R., D.P. Hougentogler, J. McGurk, E. Herrman and R.L. Alphin, 2013. Durability of incandescent, compact fluorescent and light emitting diode lamps in poultry conditions. Applied Eng. Agric., 29: 103-111.
CrossRefDirect Link - Archer, G.S., 2015. Comparison of incandescent, CFL, LED and bird level LED lighting: Growth, fear and stress. Int. J. Poult. Sci., 14: 449-455.
CrossRefDirect Link - Huth, J.C. and G.S. Archer, 2015. Comparison of two LED light bulbs to a dimmable CFL and their effects on broiler chicken growth, stress and fear. Poult. Sci., 94: 2027-2036.
CrossRefDirect Link - Rozenboim, I., B. Robinzon and A. Rosenstrauch, 1999. Effect of light source and regimen on growing broilers. Br. Poult. Sci., 40: 452-457.
CrossRefDirect Link - Zimmermann, N.G., 1988. Broiler performance when reared under various light sources. Poult. Sci., 67: 43-51.
CrossRefDirect Link - Mendes, A.S., S.J. Paixao, R. Restelatto, G.M. Morello, D.J. de Moura and J.C. Possenti, 2013. Performance and preference of broiler chickens exposed to different lighting sources. J. Applied Poult. Res., 22: 62-70.
CrossRefDirect Link - Long, H., Y. Zhao, T. Wang, Z. Ning and H. Xin, 2016. Effect of light-emitting diode vs. fluorescent lighting on laying hens in aviary hen houses: Part 1-Operational characteristics of lights and production traits of hens. Poult. Sci., 95: 1-11.
CrossRefDirect Link - Kamanli, S., I. Durmus, S. Demir and B. Tarim, 2015. Effect of different light sources on performance and egg quality traits in laying hens. Europ. Poult. Sci., 79: 1-7.
CrossRef - Min, J.K., M.S. Hossan, A. Nazma, C.N. Jae and T.B. Han et al., 2012. Effect of monochromatic light on sexual maturity, production performance and egg quality of laying hens. Avian Biol. Res., 5: 69-74.
CrossRefDirect Link - Nunes, K.C., R.G. Garcia, I.A. Naas, C. Eyng, F. Caldara, S. Sgavioli and L.G. Rambola, 2017. Iluminacao artificial com fitas de LED em substituicao a lampada fluorescente para poedeiras comerciais. Arch. Zootecn., 66: 1-5.
Direct Link - Huber-Eicher, B., A. Suter and P. Spring-Stahli, 2013. Effects of colored light-emitting diode illumination on behavior and performance of laying hens. Poult. Sci., 92: 869-873.
CrossRefDirect Link - Baxter, M., N. Joseph, V.R. Osborne and G.Y. Bedecarrats, 2014. Red light is necessary to activate the reproductive axis in chickens independently of the retina of the eye. Poult. Sci., 93: 1289-1297.
CrossRefDirect Link - Hassan, M.R., S. Sultana, H.S. Choe and K.S. Ryu, 2013. Effect of monochromatic and combined light colour on performance, blood parameters, ovarian morphology and reproductive hormones in laying hens. Ital. J. Anim. Sci., 12: 359-364.
CrossRefDirect Link - Er, D., Z. Wang, J. Cao and Y. Chen, 2007. Effect of monochromatic light on the egg quality of laying hens. J. Applied Poult. Res., 16: 605-612.
CrossRefDirect Link - Li, D., L. Zhang, M. Yang, H. Yin and H. Xu et al., 2014. The effect of monochromatic light-emitting diode light on reproductive traits of laying hens. J. Applied Poult. Res., 23: 367-375.
CrossRefDirect Link - Borille, R., R.G. Garcia, A.F.B. Royer, M.R. Santana and S. Colet et al., 2013. The use of light-emitting diodes (LED) in commercial layer production. Revista Brasileira Ciencia Avicola, 15: 135-140.
CrossRefDirect Link - Hassan, M.R., S. Sultana, H.S. Choe and K.S. Ryu, 2014. Effect of combinations of monochromatic led light color on the performance and behavior of laying hens. J. Poult. Sci., 51: 321-326.
CrossRefDirect Link - FASS., 2010. Guide for the Care and Use of Agricultural Animals in Research and Teaching. 3rd Edn., Federation of Animal Science Societies, Champaign, IL., USA.
Direct Link - Archer, G.S. and J.A. Mench, 2014. Natural incubation patterns and the effects of exposing eggs to light at various times during incubation on post-hatch fear and stress responses in broiler (meat) chickens. Applied Anim. Behav. Sci., 152: 44-51.
CrossRefDirect Link - Jones, R.B., 1986. The tonic immobility reaction of the domestic fowl: A review. Worlds Poult. Sci. Assoc. J., 42: 82-96.
CrossRefDirect Link - Archer, G.S. and J.A. Mench, 2013. The effects of light stimulation during incubation on indicators of stress susceptibility in broilers. Poult. Sci., 92: 3103-3108.
CrossRefDirect Link - Newberry, R.C. and R. Blair, 1993. Behavioral responses of broiler chickens to handling: Effects of dietary tryptophan and two lighting regimens. Poult. Sci., 72: 1237-1244.
CrossRefDirect Link








