Research Article
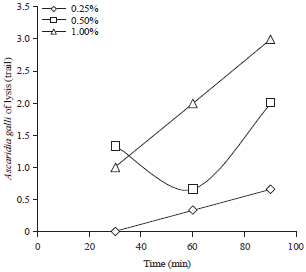
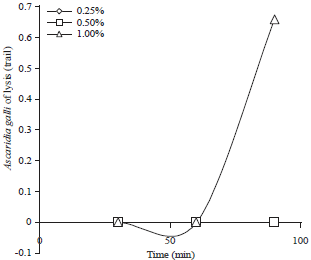
Effects of Citronella (Cymbopogon nardus) Ethanol Extracts and Distillate Dried Powder Waste on Inhibition of Ascaridia galli Development
Department of Animal Production, Agriculture Polytechnic, 85361 Kupang, East Nusa Tenggara, Indonesia
LiveDNA: 62.22572
Wihandoyo
Faculty of Animal Science, Universitas Gadjah Mada, Yogyakarta, Indonesia
LiveDNA: 62.22898
Sri Harimurti
Faculty of Animal Science, Universitas Gadjah Mada, Yogyakarta, Indonesia
R. Wisnu Nurcahyo
Faculty of Veterinary Medicine, Universitas Gadjah Mada, Yogyakarta, Indonesia