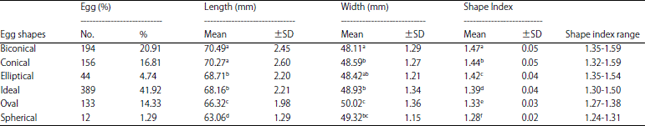
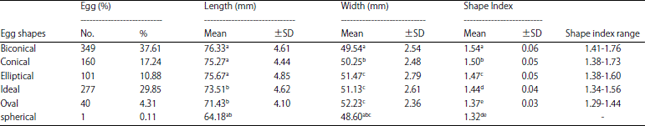
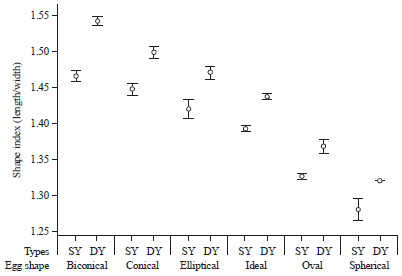
Research Article
Egg Shape is Constrained More by Width than Length, Evidence from Double-yolked Duck Eggs
School of Biology and Environmental Science, University College Dublin, Belfield, 4 Dublin, Ireland
John P. Kent
Ballyrichard House, Arklow, Co. Wicklow, Ireland