Research Article
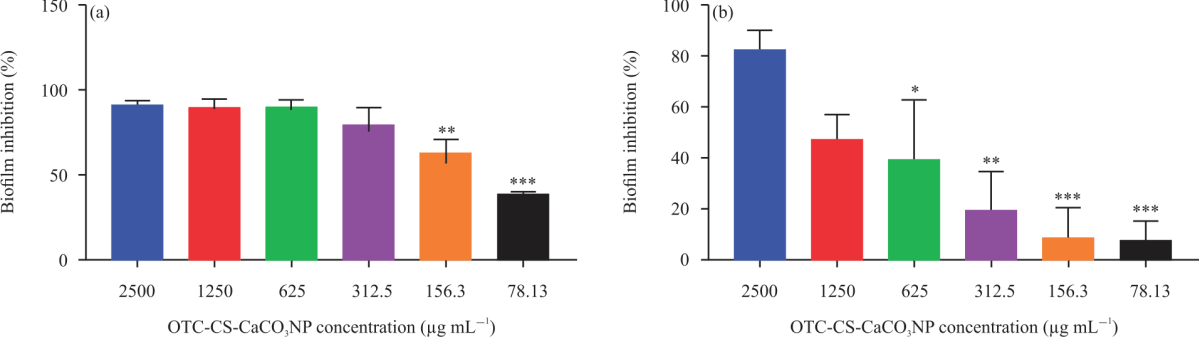
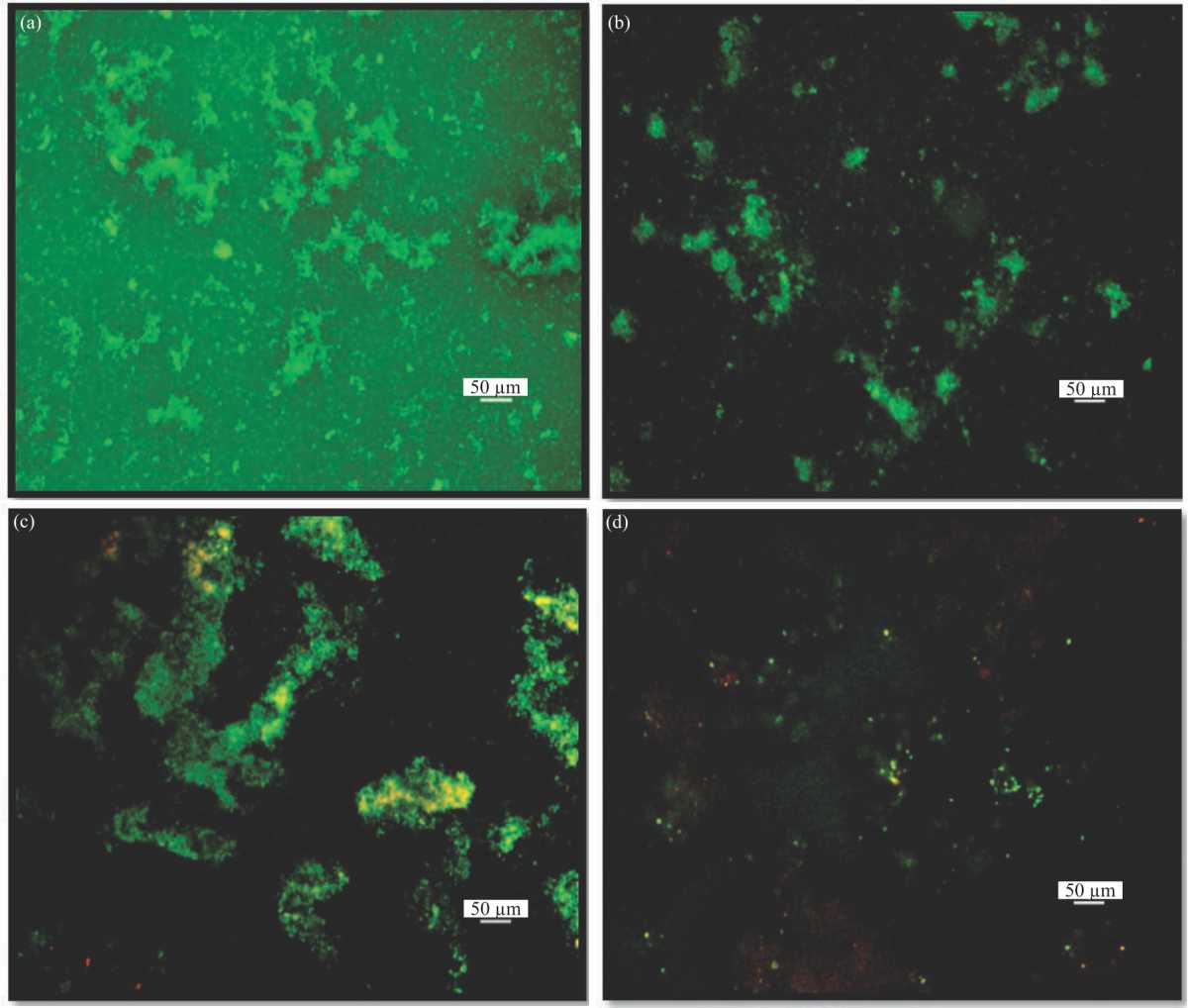
Antibiofilm Activity of Oxytetracycline Loaded Calcium Carbonate Aragonite Loaded Nanoparticle Against Corynebacterium pseudotuberculosis
Department of Veterinary Preclinical Studies, Faculty of Veterinary Medicine, Universiti Putra Malaysia, 43400 UPM, Serdang, Selangor, Malaysia
LiveDNA: 234.35358
Arifah Abdul Kadir
Department of Veterinary Preclinical Studies, Faculty of Veterinary Medicine, Universiti Putra Malaysia, 43400 UPM, Serdang, Selangor, Malaysia
LiveDNA: 60.2132
Faez Firdaus Abdullah Jesse
Department of Veterinary Clinical Studies, Faculty of Veterinary Medicine, Universiti Putra Malaysia, 43400 UPM, Serdang, Selangor, Malaysia
LiveDNA: 60.13767
Siti Zubaidah Ramanoon
Department of Farm and Exotic Animal Medicine and Surgery, Faculty of Veterinary Medicine, Universiti Putra Malaysia, 43400 UPM, Serdang, Selangor, Malaysia
Muhammad Abdul Basit
Department of Veterinary Preclinical Studies, Faculty of Veterinary Medicine, Universiti Putra Malaysia, 43400 UPM, Serdang, Selangor, Malaysia
Md Zuki Abu Bakar Zakaria
Department of Veterinary Preclinical Studies, Faculty of Veterinary Medicine, Universiti Putra Malaysia, 43400 UPM, Serdang, Selangor, Malaysia