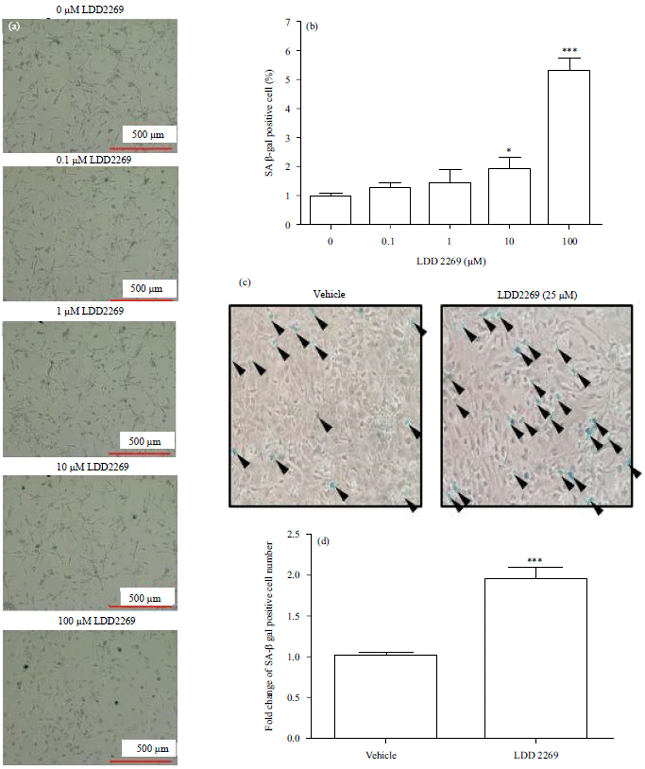
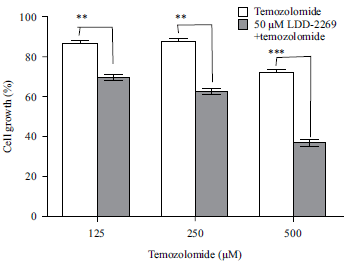
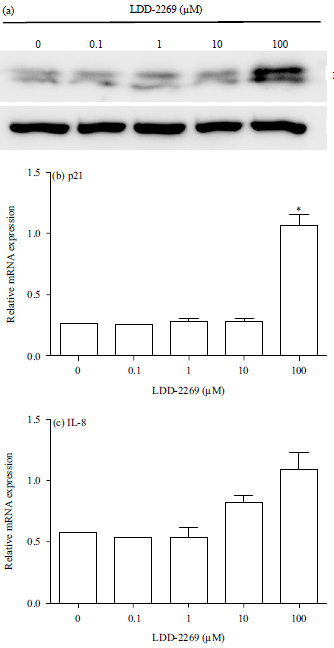Research Article
Effects of LDD-2269 on the Senescence of Glioblastoma Cells
College of Pharmacy and Research Institute of Pharmaceutical Sciences, Gyeongsang National University, Jinju-Si, Gyeongsangnam-Do 52828, Republic of Korea
Minji Bang
Department of Neuroscience, School of Medicine and Center for Neuroscience Research, Konkuk University, Seoul 05029, Republic of Korea
Chan Young Shin
Department of Neuroscience, School of Medicine and Center for Neuroscience Research, Konkuk University, Seoul 05029, Republic of Korea
Yong-Chul Kim
School of Life Sciences, Gwangju Institute of Science and Technology, Gwangju 61005, Republic of Korea
Sun-Young Han
College of Pharmacy and Research Institute of Pharmaceutical Sciences, Gyeongsang National University, Jinju-Si, Gyeongsangnam-Do 52828, Republic of Korea
LiveDNA: 82.34609