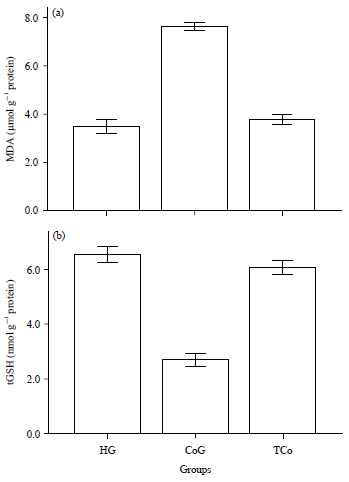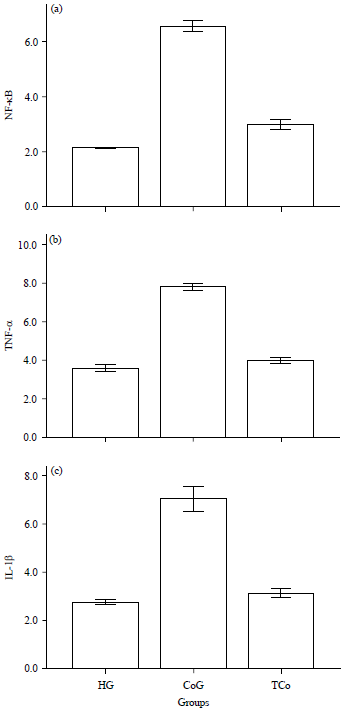Research Article
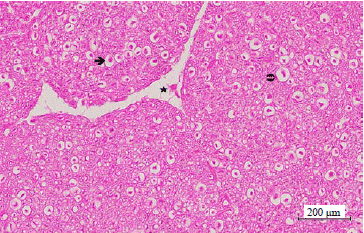
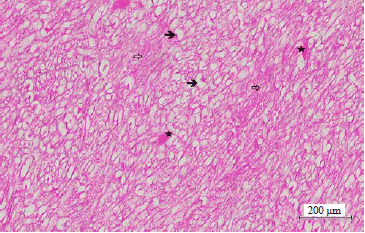
Effect of Taxifolin on Cobalt-induced Ototoxicity in Rats: A Biochemical and Histopathological Study
Department of Otorhinolaryngology, Faculty of Medicine, Meng�cek Training and Research Hospital, Erzincan Binali Yildirim University, 24100 Erzincan, Turkey
Muhammet Dilber
Dilber ENT and Aesthetic Clinic, 34158, Istanbul, Turkey
Rana Bayram
Department of Otorhinolaryngology, Faculty of Medicine, Meng�cek Training and Research Hospital, Erzincan Binali Yildirim University, 24100 Erzincan, Turkey
Esma Suleyman
Department of Otorhinolaryngology, Faculty of Medicine, Meng�cek Training and Research Hospital, Erzincan Binali Yildirim University, 24100 Erzincan, Turkey
Ertugrul Erhan
Department of Otorhinolaryngology, Faculty of Medicine, Meng�cek Training and Research Hospital, Erzincan Binali Yildirim University, 24100 Erzincan, Turkey
Sevil Karahan Yilmaz
Department of Nutrition and Dietetics, Faculty of Health Sciences, Erzincan Binali Yildirim University, 24030 Erzincan, Turkey
LiveDNA: 90.34510
Gulce Naz Yazici
Department of Histology and Embryology, Faculty of Medicine, Erzincan Binali Yildirim University, 24100 Erzincan, Turkey
Abdulkadir Coban
Department of Biochemistry, Faculty of Medicine, Erzincan Binali Yildirim University, 24100 Erzincan, Turkey
LiveDNA: 90.7968
Halis Suleyman
Department of Pharmacology, Faculty of Medicine, Erzincan Binali Yildirim University, 24100 Erzincan, Turkey
LiveDNA: 90.23571