Research Article
Effect of Luteolin and Quercetin on Thioacetamide Induced Hepatic Fibrosis in Rats
Department of Biological Sciences, Faculty of Science, University of Jeddah, Jeddah, Saudi Arabia
LiveDNA: 966.27070
The liver is important organ in metabolic stability in the body and in removal of toxins and harmful substances1. Liver diseases still record a high significant health problem worldwide that occur due to different causes that leads to oxidative stress, necrosis and inflammation of the liver cells2. Different causes may leads to liver fibrosis as chronic virus infections as hepatitis C and B, drugs, alcohol abuse, different metabolic disorders, non alcoholic steatohepatitis (NASH) and autoimmune diseases that destruct liver cells or epithelium of bile duct and end in liver fibrosis3. Regardless of the underlying cause, the mechanisms responsible for the fibrotic changes of liver cells include necrosis and apoptosis of the hepatocytes, inflammation, hepatic stellate cells (HSCs) activations, oxidative stress and excessive accumulation of extracellular-matrix (ECM) components in hepatic tissue4.
Thioacetamide (TAA) used as a fungicide drug, induced liver cirrhosis and fibrosis in different types of experimental animals5. One dose of TAA leads to centrilobular liver cell necrosis, while a chronic administrations leads to fibrosis or cirrhosis6. The TAA leads to hepatocyte destruction after its metabolism to sulfone and sulfones, sulfoxide through an important step that includes CYP4502E1-mediated biotransformation7. The treatment of liver disorders including fibrosis and cirrhosis are insufficient; no treatment successfully inhibits hepatic disease progress. Newly medications have many side effects. Therefore, it is important to find alternative medication for therapy of fibrotic hepatic disorders that are more effective and safe. There is rising interest in use of alternative medicines for liver diseases treatment. Herbs are rich in many substances as triglycerides, polyphenols and flavonoids that save the liver from damage by hepatotoxic agents8.
Luteolin (Lu)(2-(3,4-dihydroxyphenyl)-5,7-dihydroxy-4-chromenone), a bioflavonoid usually present as glycosylated forms in green pepper, celery, perilla leaf and camomile tea, etc. and as an aglycone in perilla seeds. It has diverse actions as anti-inflammatory, anti-oxidant and anti-cancer effects9 and exert anti-fibrotic effect in lung and liver10,11. Much awareness has been paid now to its anti-oxidant and anti-proliferative effects. Quercetin (Qu) (3,5,7,3,4- pentahydroxyflavone) is a popular flavonoids in human diet12 which reported to have as carcinostatic, anti-viral, anti-inflammatory, anti-fibrotic, inhibitor of aggregation of platelet and oxidation of low density lipoprotein13, cardioprotective14 and exerts anti-fibrotic actions on experimental induced allergic encephalomyelitis, airways allergic inflammatory animal model, pulmonary fibrosis and ultraviolet (UV)-irradiated primary human keratinocytes in vitro and in vivo15,16. The beneficial actions of Qu on liver destruction caused by carbon tetrachloride (CCL4) hepatotoxin or bile duct damage had been reported by Peres et al.17, Pavanato et al.18 and Li et al.19. Till now there was no successful treatment developed for liver fibrosis and from the above mentioned actions of Qu and Lu, therefore; this experimental study was designed to investigate if oral administration of Lu and Qu or their combination for three weeks exerts beneficial effects in experimental rat model of TAA-induced liver fibrosis and possible mechanisms underlying their effects.
Chemicals: Thioacetamide (C2H5NS), analytical grade was purchase from Sigma-Aldrich group, St. Louis, MO, USA. Before using TAA, it was mixed thoroughly dissolved in distilled water, according to manufacture protocol. Luteolin, commercial grade was supplied by Smart Nutrition Guillaume, Luxembourg, while Quercetin, commercial grade was supplied by I Herb, USA. Luteolin and Quercetin powders were dissolved in distilled water and mixed thoroughly. Metformin hydrochloride (Glucophage XR, 750 mg), commercial grade, was purchase from local pharmacy (Merck Serono, Darmstadt, Germany).
Animals and experimental design
Animals: Fifty four adult male Wister albino rats with an average weight 180-250 g were supplied by the animal house at King Fahd Medical Research Center (KFMRC), King Abdul-Aziz University (KAU), Jeddah, Saudi Arabia, were used in this study. The experiment was carried out for 3 months during the period from June-August, 2018. Animals were put in plastic cages (6 per cage) and left for one week in the lab before the start of the experiments for acclimatization under standard condition [humidity (65%), temperature (20±1°C) and light: dark (12/12h)]. Rats were providing with regular rodent chow and water ad libitum. King Abdelaziz University Biomedical Ethics Research Committee (Ref: 159-19) approved the study. Animal dealing was conducted according to ethical roles of the Animal Care and Use Committee of KAU and according to the international guide line of using experimental animals.
Study design: The rats were sorted randomly into 9 equal groups (n = 6):
| • | Control group (G1): It was given intraperitoneally (i.p.) saline (0.9% NaCl, vehicle) in a dose of 1 mL kg–1 b.wt., for 4 weeks |
| • | TAA group (G2): Rats were received i.p. TAA (200 mg kg–1 b.wt.) 3 times weekly for 4 weeks to induce liver fibrosis20 |
| • | Lu group (G3): Rats were given Lu via oral route (50 mg kg–1 b.wt.) daily for 3 weeks9 |
| • | Qu group (G4): Rats were received Qu via oral route (50 mg kg–1 b.wt.) daily for 3 weeks21 |
| • | Lu and Qu group (G5): Rats were received combination of Lu and Qu via oral route as groups 3 and 4 |
| • | TAA+Lu group (G6): Rats were given TAA then Lu as groups 2 and 3. TAA+Qu group (G7) rats received TAA then Qu as groups 2 and 4 |
| • | TAA+Lu+Qu group (G8): Rats received TAA then Lu and Qu in combination as groups 2 and 5 |
| • | TAA+Met group (G9): Rats received TAA (200 mg kg–1 b.wt.) 3 times weekly via i.p. route for 4 weeks then received daily i.p. injection of metformin (250 mg kg–1 b.wt.) for 3 weeks22 |
Total body weight determination: Throughout the experimental duration, animals were noticed for any behavioral or clinical abnormalities. The total body weight of all the rats were measured at start and at end of the experimental duration using a digital balance. The weight gain was calculated according to formula23:
![]()
Blood analysis: Blood samples were taken from retro-orbital veins into plain tube at the end of the experiments. About 1 mL of the blood was used for making CBC to determine total cell count of white blood cells (WBCs), red blood cells (RBCs), platelets counts (PL), hemoglobin (HBG) levels, hematocrit (HCT) and mean corpuscular volume (MCV). About 5 mL of the aliquot blood was centrifuged at 2500 rpm for 15 min and the blood serum were kept at -80°C till used. Sera samples were used to measure the levels of aspartate aminotransferase (AST), alanine aminotransferase (ALT), gamma glutamyltransferase (GGT), alkaline phosphatase (ALP), bilirubin (TBIL), total proteins (TP), albumin (ALB) and lactate dehydrogenase (LDH) using commercially available kits.
Liver weight changes: After blood sampling, the rats were scarified 24 h later after the end of the experiments by cervical dislocation after deep diethyl ether anesthesia. The abdomen was opened and livers were carefully excised, washed with saline solution, dried between filter papers and weighted. The liver index was calculated as24:
![]()
The liver was dissected into two parts, one part was homogenized and supernatant was used for assessment of oxidative stress markers and the other part of the liver was used for histological study.
Preparation of liver tissue homogenate: Tissue from right liver lobe were cut into thin slices and rinsed by cold 0.9% normal saline for further homogenization (10% w/v) in 50 mM cold potassium phosphate buffer (10% w/v). Centrifuge of homogenate was done at 3500 rpm for 10 min at 4°C. Supernatant was collected and used for assessment of oxidative stress markers.
Oxidative stress measurement in liver tissue homogenate: Malondialdehyde (MDA), superoxide dismutase (SOD), catalase (CAT) and reduced glutathione (GSH) activities in the supernatants of liver tissue homogenate were measured by using ELISA kits as described in manufacturer's steps (Nanjing Jiancheng Bioengineering Institute Co., Ltd; Nanjing, Jiangsu, China). The level of lipid peroxidation was estimated as MDA, a thiobarbituric acid reacting substance (TBARS) using 1,1,3,3, tetramethoxypropane as standard25,26. The CAT activity was measured from decomposition rate of H2O2. Reduced GSH was estimated based on formation of yellow colored complex with DTNB27. Tissue protein content was measured using bovine serum albumin (BSA) as a standard28.
Histological examinations of the liver: For liver histopathological examination, midsections of left liver lobes were processed for light microscopy. This processing consisted of fixing the specimen in a 5% neutral formalin solution, dehydrated in gradual ethanol (50-100%), cleared in xylene and then embedding in paraffin, slicing sections 5 mm in thickness and staining with hematoxylin and eosin to examined microscopically.
Statistical analysis: All of the values are expressed as Mean±Standard error of means (SEM). Data was analyzed using Statistical Package for Social Sciences (SPSS Inc., version 22, Chicago, IL, USA). The data were analyzed using one-way ANOVA test followed by least significant difference (LSD) test for comparison with control and hepatotoxin TAA groups. A p≤0.05 was statistically significant.
Effects of TAA, Lu and Qu on total body weight and liver weight: Final body weight and percentage changes in body weight were significantly higher in Lu, Qu, Lu+Qu, TAA+Qu, TAA+Lu+Qu and TAA+Metformin groups versus control and TAA groups (Table 1). Rats received TAA for 4 weeks showed significant increase in liver weight index versus control. Meanwhile, liver index was significantly decreased in Lu, Qu, Lu+Qu, TAA+Lu, TAA+Qu, TAA+Lu+Qu and TAA+Metformin groups versus TAA groups (Table 2).
Effect of TAA, Lu, Qu administration on complete blood count: The TAA administration led to significant elevation of WBCs count but significant decrease in RBCs, platelets counts and HBG level versus control. Administration of Lu or Qu led to significant decrease in WBCs count, but significant increase in HBG, HCT, MCV, RBCs and platelets counts versus TAA treated group. In TAA+Lu, TAA+Qu and TAA+Lu+Qu groups, WBCs count was significantly decreased, but RBCs count, HBG and HCT were significantly increased versus TAA treated group. Treatment of the rats with TAA and metformin led to significant decrease in WBCs count, but significant increase in RBCs count, HBG and platelets count versus TAA treated group. Meanwhile, WBCs count was still significantly higher than control (Table 3).
Effect of TAA, Lu, Qu administration on liver functions: Administration of TAA led to significant elevation in the serum levels of AST, ALP, ALT, GGT, LDH and bilirubin but significant decreased in total proteins and albumin versus to control. Treatment of the rats with Lu or Qu or their combination or metformin after TAA administration led to improvement of liver function versus TAA group but still impaired compared with control (Table 4).
Effect of TAA, Lu, Qu administration on oxidative stress: Administration of TAA to the rats led to significant increase in liver tissue homogenate levels of MDA, but significant decline in CAT, SOD and GSH. Administration of Lu and Qu or their combination or metformin led to significant decrease in MDA, but significant increase in CAT, SOD and GSH compared to TAA alone treated group but these parameters were still changed versus control group (Table 5).
Histological examination of the liver: Liver section examination revealed normal hepatocytes radiating from the central vein (CV) and separated by thin blood sinusoids in the control group. In TAA group, hepatocyte necrosis around the central and portal veins (PV) were found associated with marked fibroblast proliferation and collagen fiber formation bridging the two regions leading to prominent liver lobulation. Liver of Lu, Qu and Lu and Qu groups showed normal hepatocytes separated by blood sinusoids without any changes compared to control.
| Table 1: | Comparison of body weight in different studied groups |
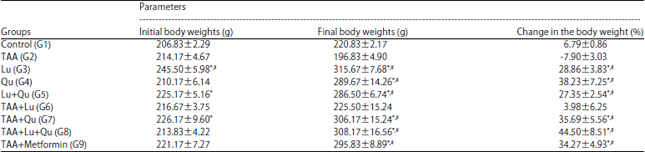 | |
Data expressed as Mean±Standard error, *Significance versus control (G1), #Significance versus TAA (G2), Significance was made using one way ANOVA (LSD) test | |
| Table 2: | Comparison of liver weight and liver index in different studied groups |
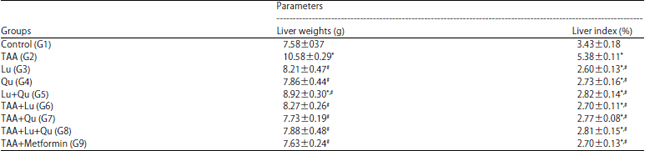 | |
| Data expressed as Mean±standard error, *Significance versus control (G1), #Significance versus TAA (G2), Significance was made using one way ANOVA (LSD) test | |
| Table 3: | Comparison of hematological changes in different studied groups |
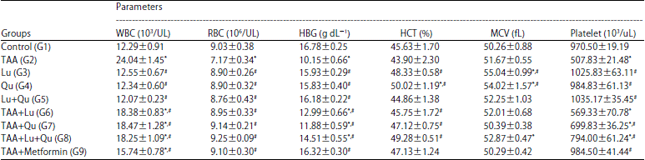 | |
Data expressed as Mean±Standard error, *Significance versus control (G1), #Significance versus TAA (G2), Significance was made using one way, ANOVA (LSD) test, WBCs: White blood cells, RBCs: Red blood cells, HBG: Hemoglobin, HCT: Hematocrit, MCV: Mean corpuscular volume | |
| Table 4: | Comparison of liver function tests in different studied groups |
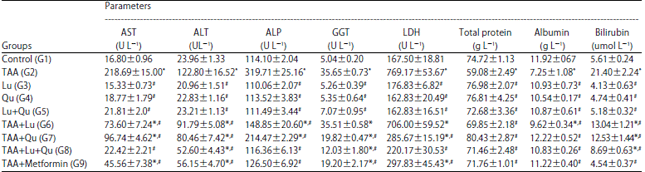 | |
Data expressed as Mean±Standard error, *Significance versus control (G1), # Significance versus TAA (G2), Significance was made using one way ANOVA (LSD) test, AST: Aspartate aminotransferase, ALT: Alanine aminotransferase, ALP: Alkaline phosphatase, GGT: Gamma glutamyl transferase, LDH: Lactate dehydrogenase | |
| Table 5: | Comparison of oxidative stress markers in liver tissue homogenate in different studied groups |
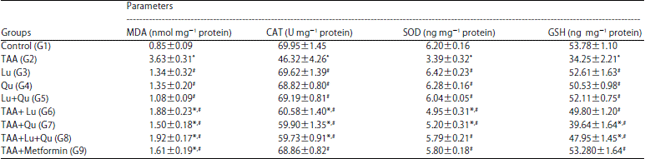 | |
Data expressed as Mean±Standard error, *Significance versus control (G1), #Significance versus TAA (G2), Significance was made using one way ANOVA (LSD) test, MDA: Malondialdehyde, CAT: Catalase, SOD: Superoxide dismutase, GSH: Reduced glutathione | |
Liver of TAA+Lu showed marked protection against TAA induced centro-lobular necrosis. Few lymphocyte cell infiltrate could be seen near CV. Liver of TAA+Qu group showed absence of necrotic observed in TAA group, but showed residual slight bridging and lymphocyte sinusoidal infiltration. Liver of TAA+Lu and Qu group showed no fibrosis or bridging but slight congestion was observed. Liver of ,TAA+Met group showed nearly normal hepatocytes, residual necrosis and cell infiltration with less fibrosis (Fig. 1G1-G9).
Liver fibrosis is a chronic wound healing after liver damage29. This study showed that the maximum increases in total body weight were noticed in control rats which representative normal growth. The minimum increases in total body weight were found in TAA treated rats30 that may be due to enhanced lipolysis21, decrease appetite31, reduced nutrients absorption, increased protein catabolism, decreased of food utilization or excessive water, salts and proteins loss due to renal injury6.
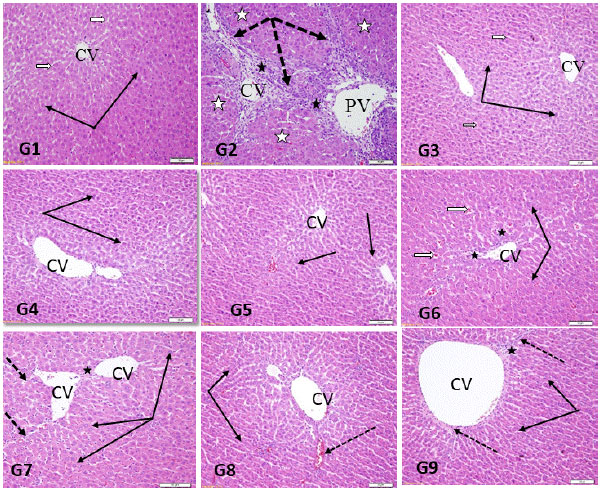 | |
| Fig. 1(G1-G9): | Sections in rat’s liver stained by H and E showing central vein (CV) region of, G1 (Control group): Normal hepatocytes (thin black arrows) arranged in normal radial pattern around CV with normal hepatic sinusoids (white arrows), G2 (TAA group): Marked fibrocollagenous tissue proliferation extending from portal area forming porto-portal bridging fibrosis (dotted arrows) with intralobular fibrous septa dividing hepatocytes (white stars), G3 (Lu group): Normal hepatocytes (thin black arrows) with normal hepatic sinusoids (white arrows) G4 (Qu group): Normal hepatocyte (black arrows) and normal portal area, G5 (Lu and Qu group): Normal hepatocyte (thin black arrows) with no changes compared to control, G6 (TAA+Lu group): Mild lymphocytic infiltration (black stars) around central vein seen with dilatation of hepatic sinusoids (white arrow) and normal hepatocytes (black arrow), G7 (TAA+Qu group): Mild intralobular fibroblastic proliferation (dotted arrow) with normal hepatocytes (black arrow) with normal portal area (star), G8 (TAA+Lu and Qu group): Normal hepatocytes (thin black arrows) with normal hepatic sinusoids (black arrow), G9: Normal hepatocytes (thin black arrows), residual necrosis (black star) with less fibrosis (dotted arrow) |
The liver weight was significantly increased in TAA treated rats3,32 that could be explained by inflammatory changes seen in the liver as vascular congestion, leukocyte infiltrations and fibrosis. Meanwhile, TAA- groups treated with Lu or Qu or their combination reversed those defects. The hepatoprotective activity of Lu in acute liver injury in mice was reported previously10,33. Qu inhibits liver fibrosis by direct downregulation of HMGB1-TLR2/4-NFκB signaling pathway19, suppression of HSC activation and reducing autophagy through regulating crosstalk between TGF-β1/Smads and PI3K/Akt signals34.
TAA treatment led to significant decrease in RBCs, HGB, HCT and MCV, but significant increase in WBCs count. The TAA administration leads to inflammation that increased WBCs count and the toxic effects of TAA leads to decrease in RBCs count and HGB content35. Lu and Qu treatment significantly reversed TAA-induced alteration of CBC.
TAA intake induced serum elevation of hepatic specific enzymes due to damage of cell membrane leads to liberates of these enzymes into circulation36-38. Elevated bilirubin levels may be due to blockage of bile ductile caused by inflammation and fibrosis in portal triads and/or regurgitation of conjugated bilirubin from necrotic hepatocytes to sinusoids39. Co-treatments with Lu and Qu in TAA-treated rats significantly decreased ALT and AST levels than TAA-treated rats. It was reported that Qu decreases serum levels of ALT, ALP, AST and LDH18,40.
TAA administration led to significant increase of MDA, but significant decreased of CAT, SOD and GSH in liver tissue homogenate than control group. The TAA contains a thiono-sulfur group which undergoes extensive metabolism to produce acetamide and TAA-S-oxide, that further transformed into TAA-S, S-dioxide, unstable reactive metabolites that initiate necrosis by covalently binding to liver macromolecules41. Increased oxidative stress could result also from activated neutrophils, macrophage and monocytes which release various pro-oxidants42. Current results revealed that administration of Lu and Qu to TAA induced liver fibrosis in rats significantly alleviated the TAA-suppressive effect on antioxidant enzymes SOD, GSH and CAT. Hepatic ROS production was significantly attenuated by Lu administration in both Cx32ΔTg and Wt rats43. Rats orally treated with Qu for 3 weeks after induction of hepatotoxicity showed signs of improvement by histological and biochemical studies21. Qu administration orally (10 mg kg–1 daily) for 4 weeks reduce dimethylnitrosamine-induced liver injury and increase serum albumin and liver GSH levels and decrease lipid peroxidation44. Administration of Qu (500 μL kg–1 i.p.) for 3 weeks was able to restore GSH, SOD and CAT levels in CCL4 induced liver cirrhosis in rats3. Qu decreased histological signs of acute inflammation in the treated animals via suppressing leucocyte recruitment, decreasing chemokine levels and MDA and increasing antioxidant enzyme activity in experimental rat model45.
In this work, TAA administration resulted in a marked necrosis of hepatocytes, fibroblasts proliferation and lymphocyte infiltration. Upon liver injury, hepatic stellate cells acquire a highly proliferative index producing fibrillar collagen within the injured liver46. Lu administration potentially protected against TAA induced liver histological changes in rat. The anti- fibrotic effect of Lu in liver fibrosis was reported47. Lu protected human skin fibroblasts against oxidative stress induced by ultra violet radiation exposure48. Lu deactivated hepatic stellate cells that enhance fibrosis10. Lu protected rat liver from acetaminophen-induced hepatotoxicity49. In the present study, Qu provided marked protection against TAA-induced hepatic histological changes. Qu prevented hepatic fibrosis induced by CCl4 in mice through inhibition of stellate cell inactivation34. A combination of Lu and Qu result in optimum significant protection of rat hepatic tissue against TAA induced necrotic changes50.
The results of the present study indicated that the both Qu and Lu showed the hepatoprotective effect against TAA induced biochemical and histological changes in rat liver. Further pharmacological evidences at molecular level are required to establish the mechanism of the action of these natural substances.
The present study discovered for the first time that using of both Qu and Lu resulted in synergetic effect of their individual hepatoprotective effects against TAA induced changes. These finding could be beneficial and need further pharmacological researches at molecular level to establish further mechanism of the action and optimum given doses and duration of these natural substances.