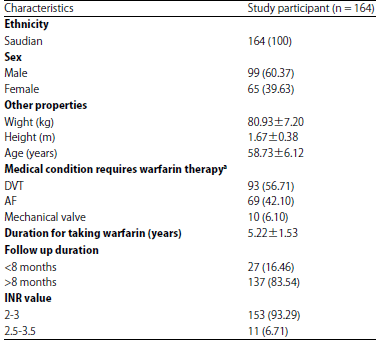
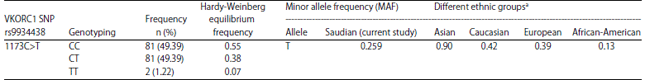
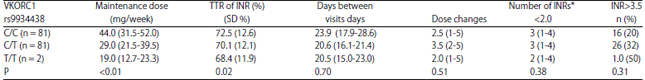
Research Article
Influence of Vitamin K Epoxide Reductase Complex 1 Polymorphism on Warfarin Therapy in a Cohort Study of Saudi Patients
Department of Clinical Pharmacy, College of Pharmacy, Prince Sattam Bin Abdulaziz University, 11942 Al-Kharj, Saudi Arabia
LiveDNA: 966.20248
Mohamed Abd-Elghany Abd-Elaziz
Department of Clinical Pharmacology, Mansoura Faculty of Medicine, Mansoura University, 35516 Mansoura, Egypt
Rehab Hamdy Ashour
Department of Clinical Pharmacology, Mansoura Faculty of Medicine, Mansoura University, 35516 Mansoura, Egypt
Taimour Langaee
Department of Pharmacotherapy and Translational Research at the University of Florida (UF), College of Pharmacy, Gainesville, FL 32610, USA