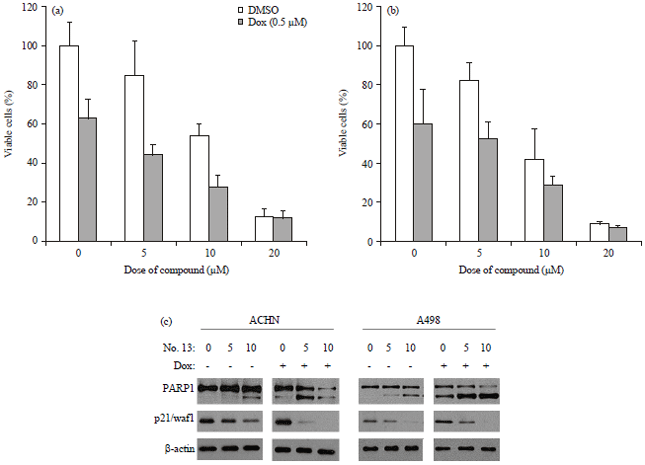
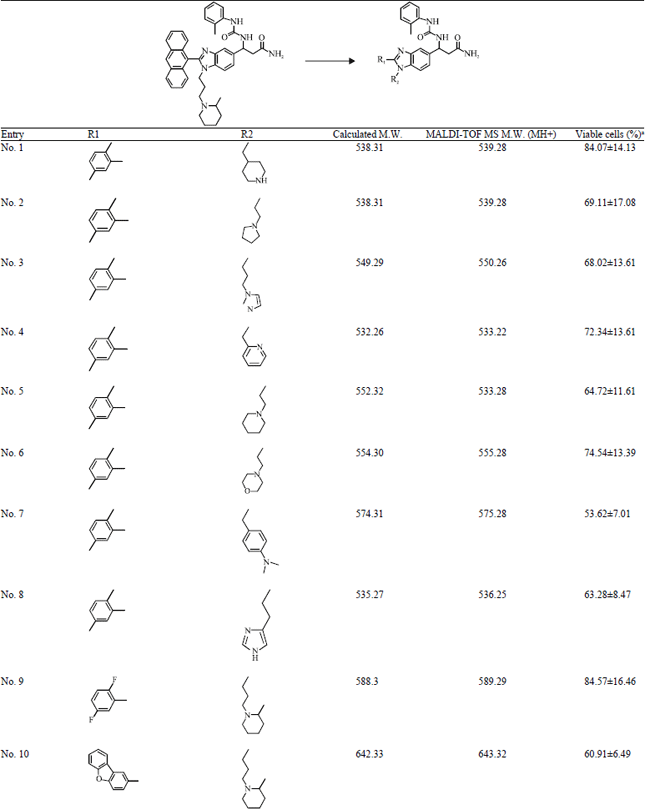
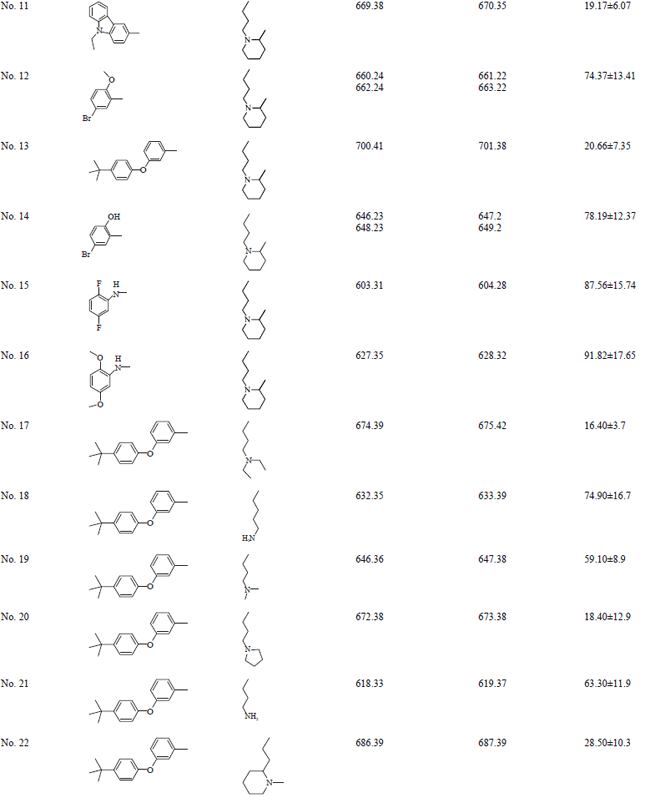
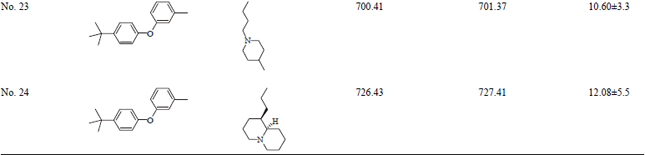
Short Communication
Structure-Activity-Relationship Study of the Novel p21/waf1 Inhibitor for Anti-Cancer Agents against Renal Cell Carcinoma
Program in Nano Science and Technology, Department of Transdisciplinary Studies, Seoul National University Graduate School of Convergence Science and Technology, Suwon, 443-270, South Korea