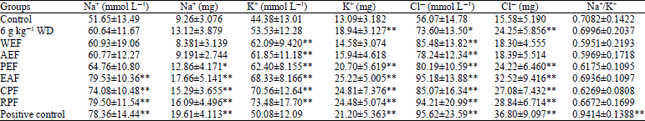
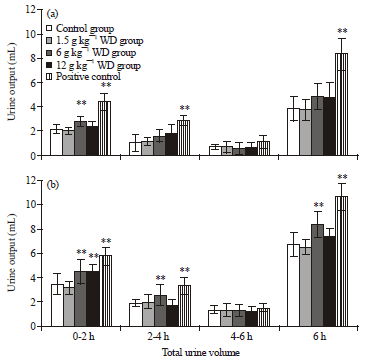
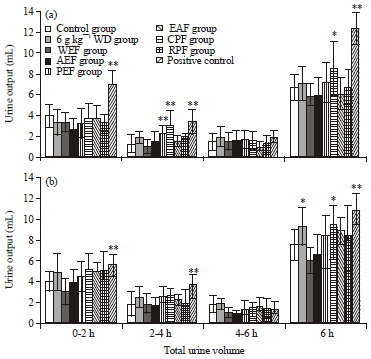
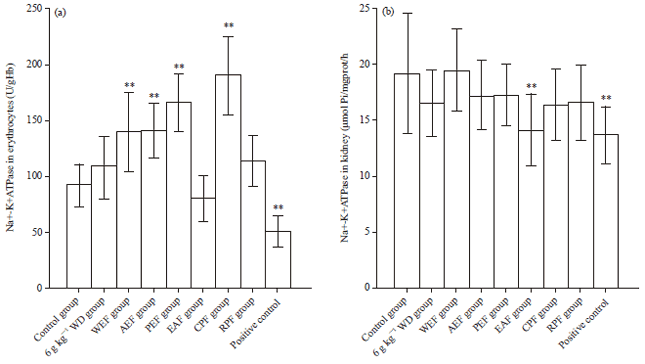
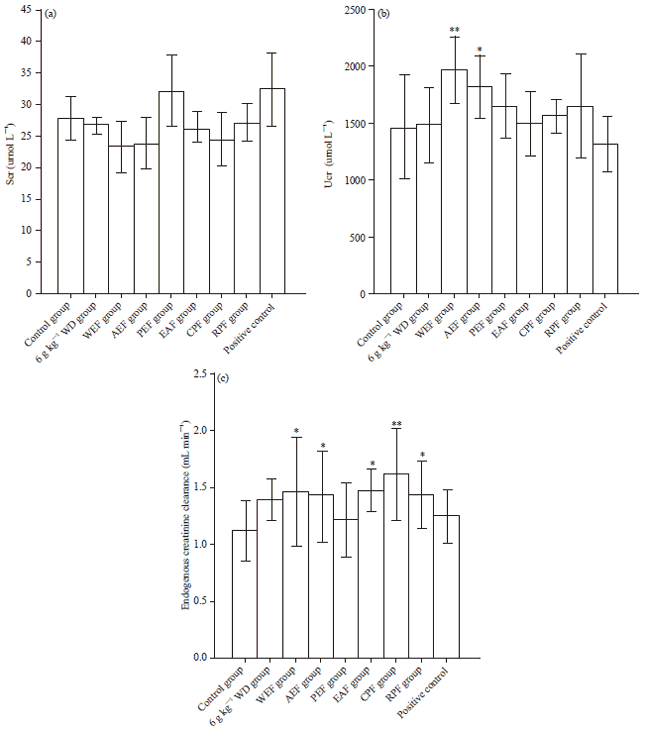
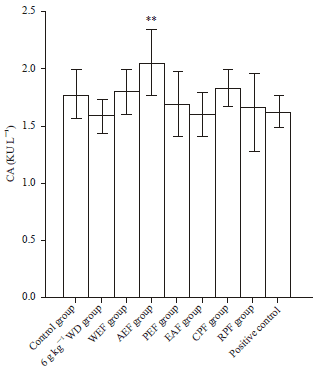
Research Article
Diuretic Ingredients of Poria coco
College of Pharmacy, Liaoning University of Traditional Chinese Medicine, 77 Life One Road, DD Port, Dalian, 116600, China
Ding Yan-Xia
College of Pharmacy, Liaoning University of Traditional Chinese Medicine, 77 Life One Road, DD Port, Dalian, 116600, China
Dou De-Qiang
College of Pharmacy, Liaoning University of Traditional Chinese Medicine, 77 Life One Road, DD Port, Dalian, 116600, China
Ran Xiao-Ku
College of Pharmacy, Liaoning University of Traditional Chinese Medicine, 77 Life One Road, DD Port, Dalian, 116600, China
Xu Yu-Bin
College of Pharmacy, Liaoning University of Traditional Chinese Medicine, 77 Life One Road, DD Port, Dalian, 116600, China
Li Ling-Hui
College of Pharmacy, Liaoning University of Traditional Chinese Medicine, 77 Life One Road, DD Port, Dalian, 116600, China
Kang Ting-Guo
College of Pharmacy, Liaoning University of Traditional Chinese Medicine, 77 Life One Road, DD Port, Dalian, 116600, China