Research Article
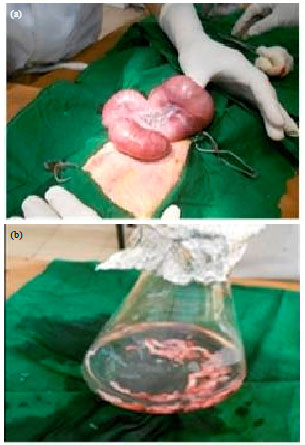
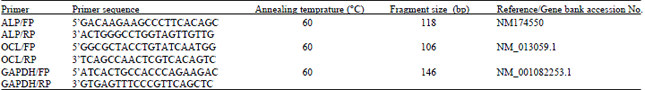
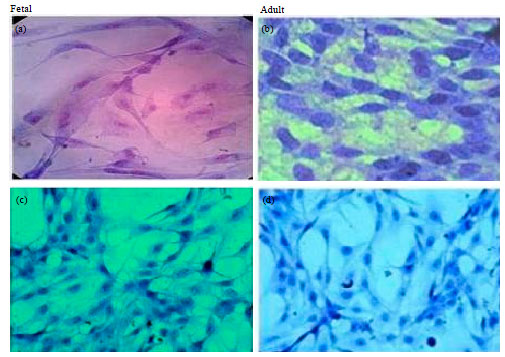
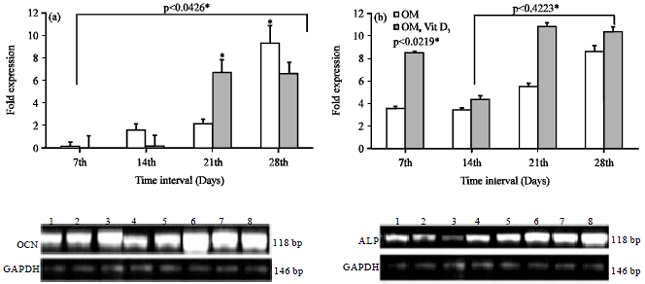
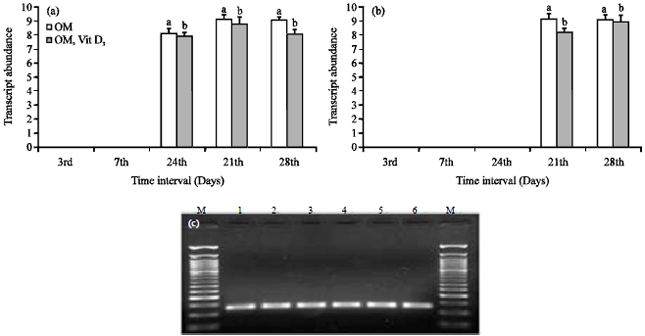
Evaluation of in vitro Efficacy of Vitamin D3 on the Osteogenic Differentiation and Mineralization Capabilities of Fetal and Adult Osteoblasts of Rabbit Reflects Therapeutic Potential
Division of Surgery,Indian Veterinary Research Institute, Izatnagar, Bareilly (UP), 243122, India
Amarpal
Division of Surgery,Indian Veterinary Research Institute, Izatnagar, Bareilly (UP), 243122, India
P. Tamilmahan
Division of Surgery,Indian Veterinary Research Institute, Izatnagar, Bareilly (UP), 243122, India
Kuldeep Dhama
Division of Pathology, Indian Veterinary Research Institute, Izatnagar, Bareilly (UP), 243122, India
Netrapal Sharma
Division of Surgery,Indian Veterinary Research Institute, Izatnagar, Bareilly (UP), 243122, India