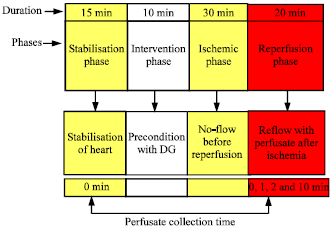
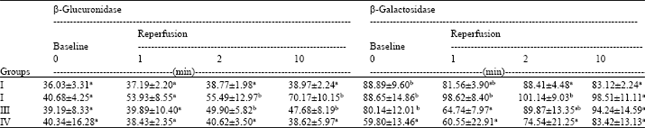
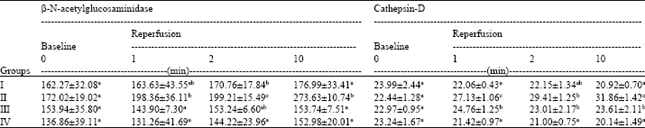
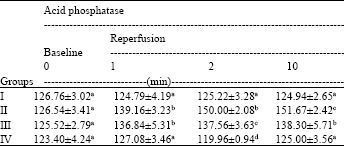
Research Article
Effect of Myocardial Ischemic Reperfusion Injury on Lysosomal Enzymes and Electrolytes Content of Perfusate and Cardiac Tissue under the Influence of Desmodium gangeticum L.: An In-vitro Study
Department of Pharmacology, Amrita School of Pharmacy, Amrita Institute of Medical Science, Amrita Vishwa Vidyapeetham University, Kochi-682041, Kerala, India
L. Upadhyay
Advance Research Centre for Indian System of Medicine, PRIST University, Thanjavur-613 403, Tamil Nadu, India