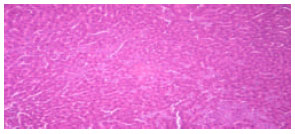
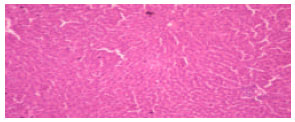
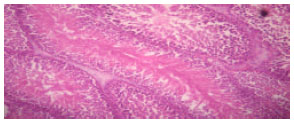
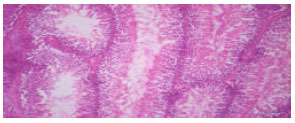
Research Article
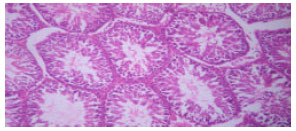
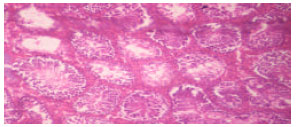
Biochemical and Histological Responses of Hepatotoxic Rats Fed Musa paradisiaca L. Supplemented Diet
Biochemistry and Molecular Biology Unit, Department of Biological Sciences, Covenant University, P.M.B 1023, Ota, Ogun State, Nigeria
I. C. Obichi
Biochemistry and Molecular Biology Unit, Department of Biological Sciences, Covenant University, P.M.B 1023, Ota, Ogun State, Nigeria
O. E. Omotosho
Biochemistry and Molecular Biology Unit, Department of Biological Sciences, Covenant University, P.M.B 1023, Ota, Ogun State, Nigeria