Research Article
Biochemical, Haematological and Histological Effects of Dietary Supplementation with Leaves of Gnetum africanum Welw. on Paracetamol-induced Hepatotoxicity in Rats
Biochemistry and Molecular Biology Unit, Department of Biological Sciences,Covenant University, P.M.B. 1023, Ota, Ogun State, Nigeria
Atinuke O. Osundiya
Biochemistry and Molecular Biology Unit, Department of Biological Sciences,Covenant University, P.M.B. 1023, Ota, Ogun State, Nigeria










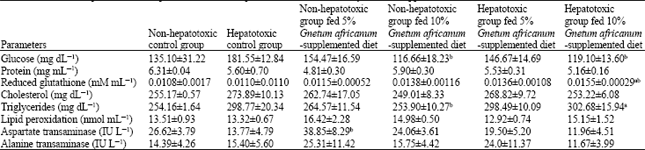

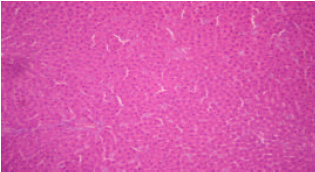
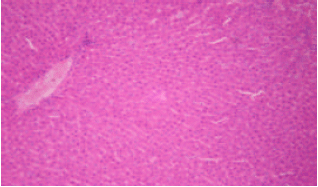
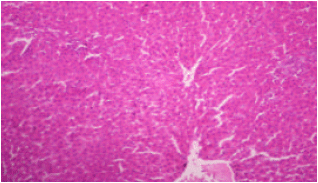
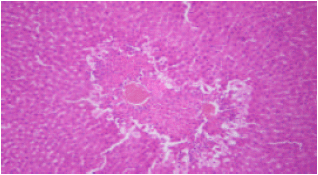
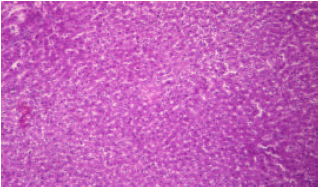
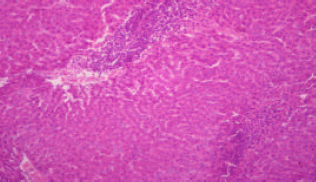
sankar anand R Reply
superb work its so useful to me as i am an pharmacologist
sone enone bertin Reply
very interresting study
it is aqueous or alcohol crude extract?
secondly what about male rate?
i am investigated in the same subject but using female and male rats, G. africanum and G. buchholzianum
thanks
sone enone bertin Reply
how can i get a full text pleased
sone enone bertin Reply
how can i connect to the authors?