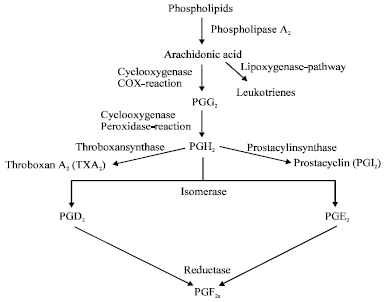
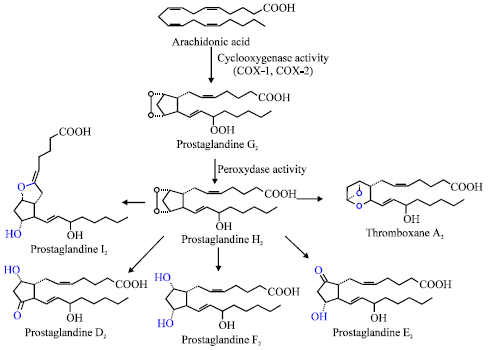
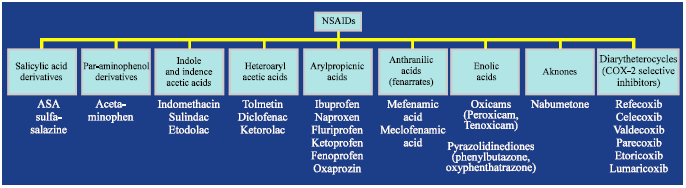
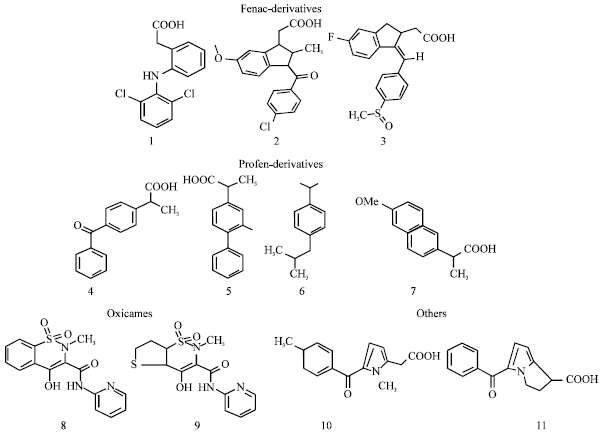
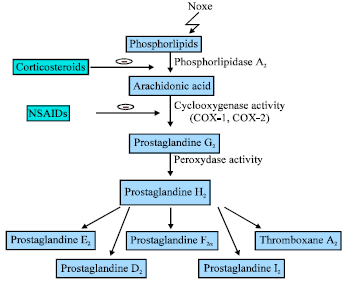
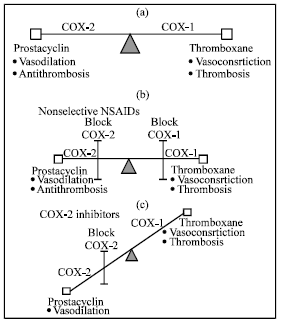
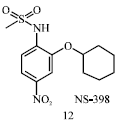
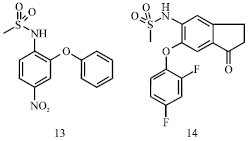
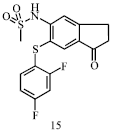
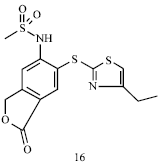
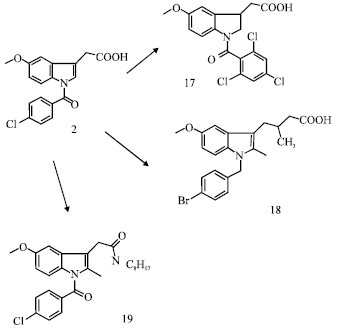
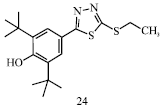
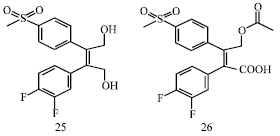
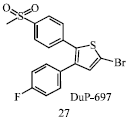
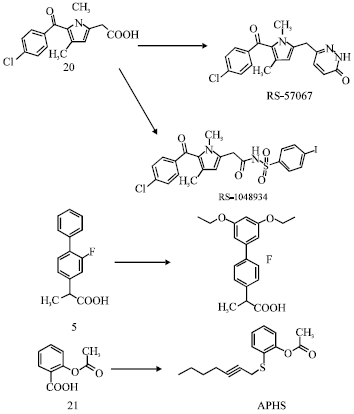
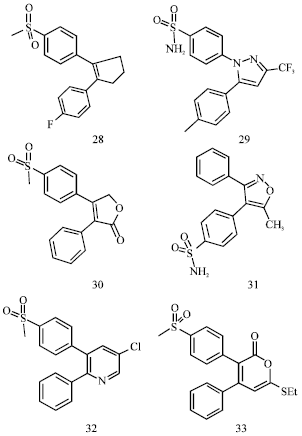
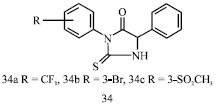
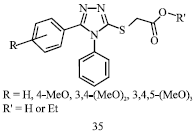
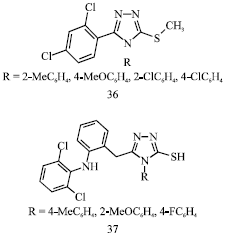
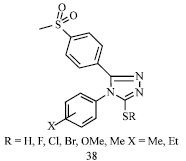
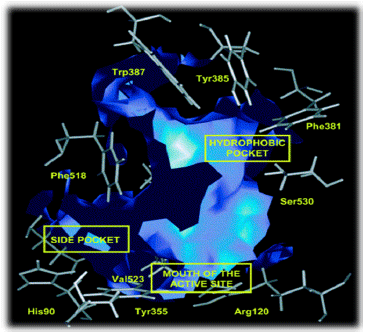
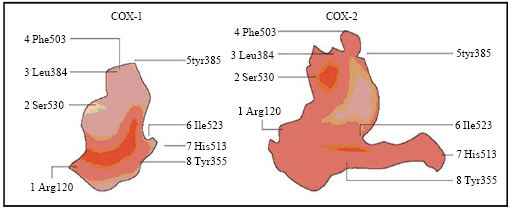
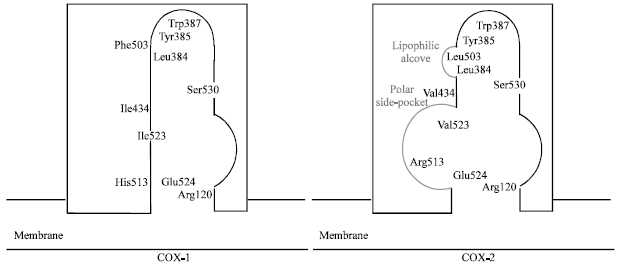
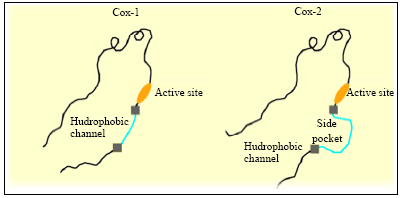
Review Article
Therapeutic and Toxic Effects of New NSAIDs and Related Compounds: A Review and Prospective Study
Department of Pharmaceutical Chemistry, College of Pharmacy, King Saud University, 11451 Riyadh, Saudi Arabia
L. A. Abou-Zeid
Department of Pharmaceutical Organic Chemistry, Faculty of Pharmacy, University of Mansoura, Mansoura 35516, Egypt
I. A. Shehata
Department of Pharmaceutical Chemistry, College of Pharmacy, King Saud University, 11451 Riyadh, Saudi Arabia
M. A. Al-Omar
Department of Pharmaceutical Chemistry, College of Pharmacy, King Saud University, 11451 Riyadh, Saudi Arabia