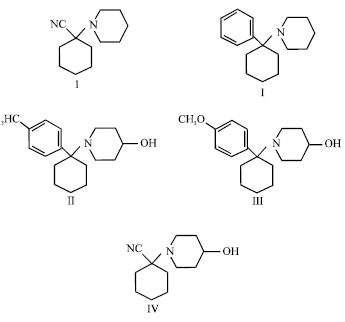
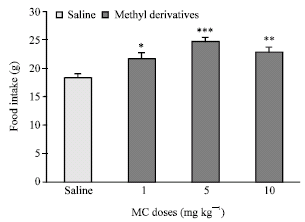
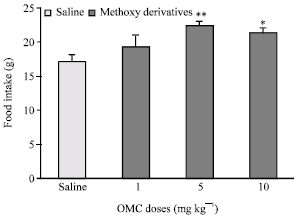
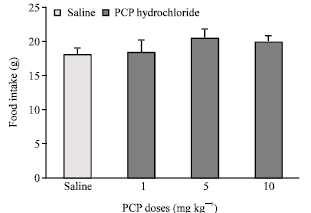
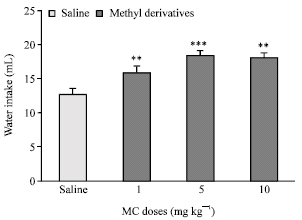
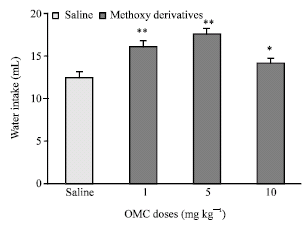
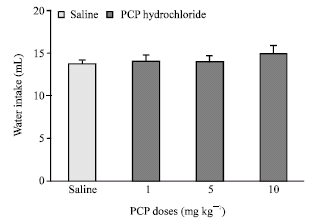
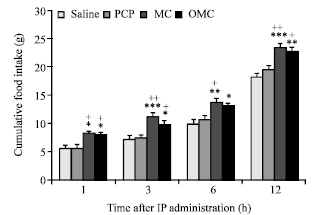
Research Article
Effects of Methyl and Methoxy Derivatives of Phencyclidine on Food and Water Intake
Department of Biology, Club, Islamic Azad University, Karaj Branch, Karaj, Iran
A. Ahmadi
Department of Chemistry, Club, Islamic Azad University, Karaj Branch, Karaj, Iran
A.A. Salari
Young Researchers Club, Islamic Azad University, Karaj Branch, Karaj, Iran