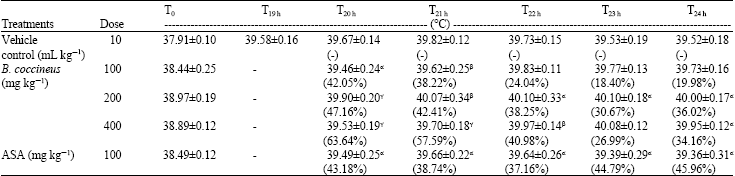
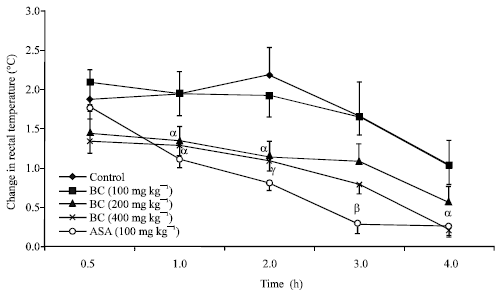
Research Article
Antipyretic Activity of Byrsocarpus coccineus Schum and Thonn. (Connaraceae)
Department of Pharmacology, College of Medicine, University of Lagos, P.M.B. 12003, Lagos, Nigeria
O. O. Adeyemi
Department of Pharmacology, College of Medicine, University of Lagos, P.M.B. 12003, Lagos, Nigeria