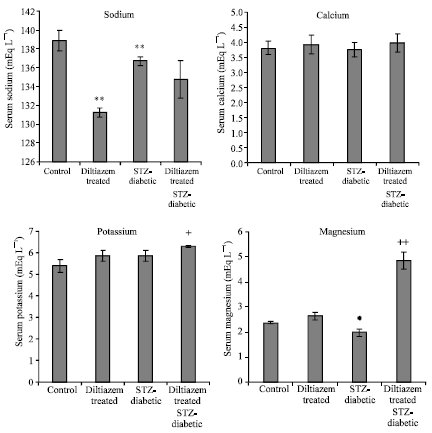
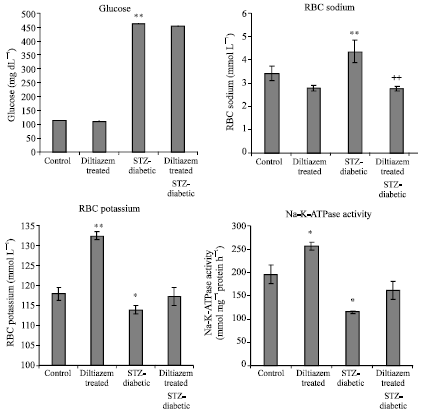
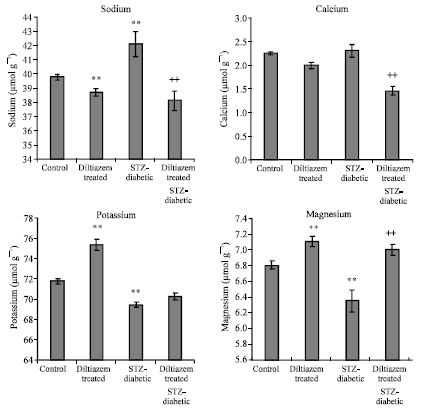
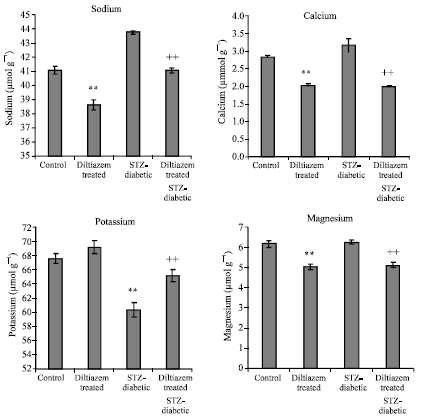
Research Article
Effects of Diltiazem on Electrolytes Homeostasis in Streptozotocin-Induced Diabetic Rats
Department of Biochemistry, University of Karachi, Karachi-75270, Pakistan
Syed Muhammad Shahid
Department of Biochemistry, University of Karachi, Karachi-75270, Pakistan
Tabassum Mahboob
Department of Biochemistry, University of Karachi, Karachi-75270, Pakistan