Research Article
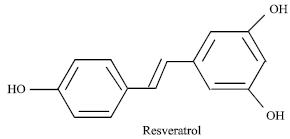
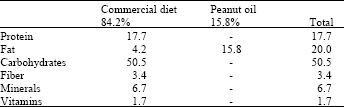
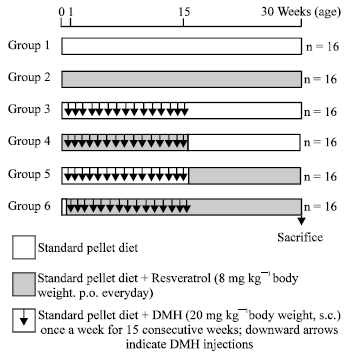
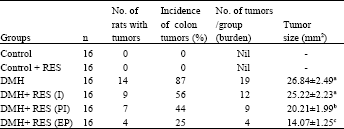
Resveratrol, a Phytoalexin Enhances Hepatic Antioxidant Defense in 1,2-dimethylhydrazine-induced Colon Carcinogenesis
Department of Biochemistry and Biotechnology, Faculty of Science, Annamalai University, Annamalainagar-608 002, Tamilnadu, India
Namasivayam Nalini
Department of Biochemistry and Biotechnology, Faculty of Science, Annamalai University, Annamalainagar-608 002, Tamilnadu, India