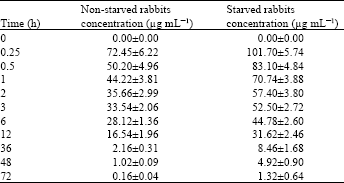
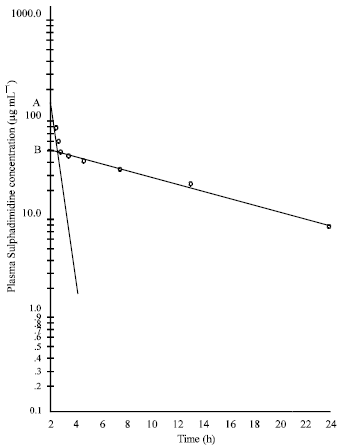
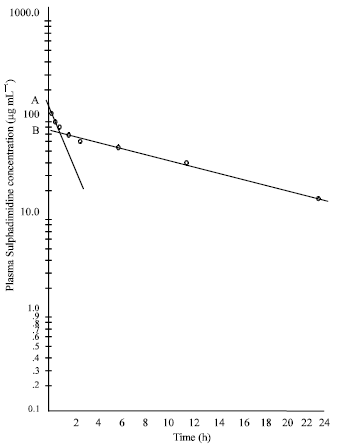
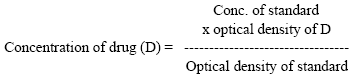Research Article
The Effect of Short Term Starvation on the Plasma Kinetics of Sulphadimidine in Rabbits
Department of Pharmacology, Usmanu Danfodiyo University, Sokoto, Nigeria
A. M. Umarudeen
Department of Pharmacology, Usmanu Danfodiyo University, Sokoto, Nigeria
P. A. Onyeyili
Department of Veterinary Pharmacology and Physiology, University of Maiduguri, Nigeria
A. T. Elsa
Department of Pharmacology, Usmanu Danfodiyo University, Sokoto, Nigeria