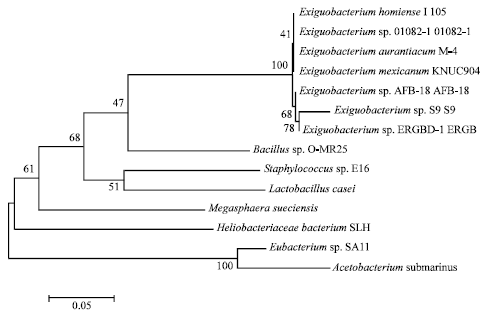
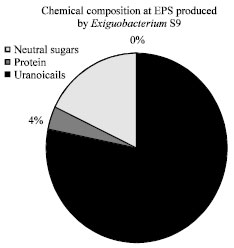
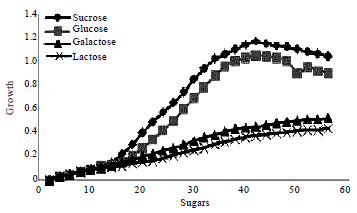
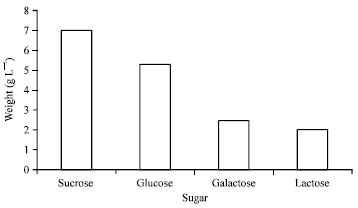
Research Article
Isolation and Characterization of Exopolysaccharide Producing Bacteria from Pak Bay (Mandapam)
Department of Microbiology, Hajee Karutha Rowther Howdia College, Uthamapalayam 625533, Tamilnadu, India
Vignesh Venkatasamy
Department of Animal Science, School of Life Sciences, Bharathidasan University, Tiruchirapalli 620 024, Tamilnadu, India
Thirumurugan Ramasamy
Department of Animal Science, School of Life Sciences, Bharathidasan University, Tiruchirapalli 620 024, Tamilnadu, India