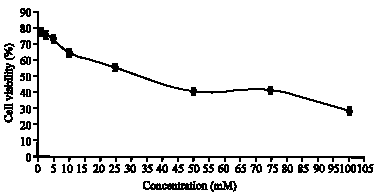
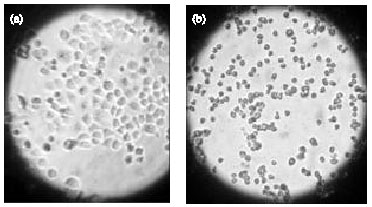
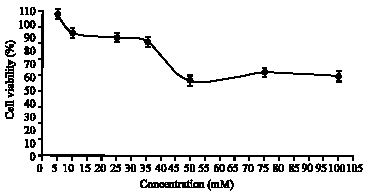
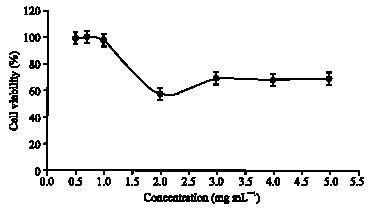
Research Article
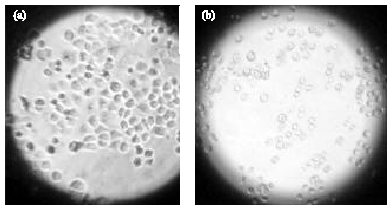
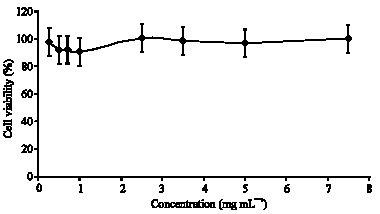
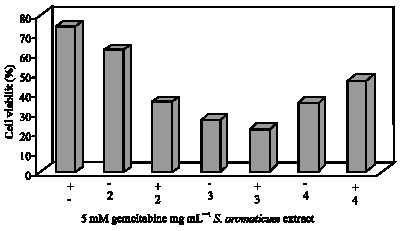
Clove (Syzygium aromaticum) Extract Potentiates Gemcitabine Cytotoxic Effect on Human Cervical Cancer Cell Line
Department of Biotechnology, Manipal University, Dubai, UAE
S. Sasidharan
Department of Biotechnology, Manipal University, Dubai, UAE
T. Ahmed
Department of Biotechnology, Manipal University, Dubai, UAE
M. Ahmed
Department of Biotechnology, Manipal University, Dubai, UAE
C. Sharma
Department of Biotechnology, Manipal University, Dubai, UAE