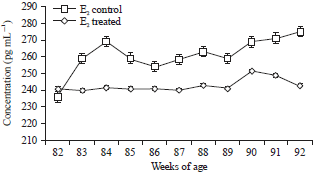
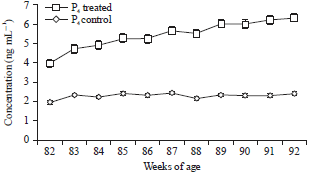
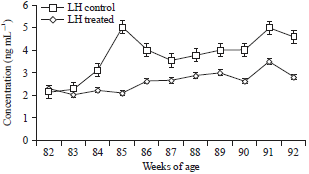
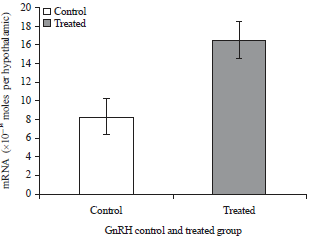
Research Article
Endocrine Changes in Hens (Gallus gallus domesticus) Exposed to Red Spectrum of Light
Division of Animal Physiology, National Institute of Animal Nutrition and Physiology, Adugodi, Hosur Road, 560 030 Bangalore, India
Ashish Mishra
Division of Animal Physiology, National Institute of Animal Nutrition and Physiology, Adugodi, Hosur Road, 560 030 Bangalore, India
S. Mondal
Division of Animal Physiology, National Institute of Animal Nutrition and Physiology, Adugodi, Hosur Road, 560 030 Bangalore, India
LiveDNA: 91.1478
Vaibhav Awachat
Division of Animal Physiology, National Institute of Animal Nutrition and Physiology, Adugodi, Hosur Road, 560 030 Bangalore, India
G. Ravi Kiran
Division of Animal Physiology, National Institute of Animal Nutrition and Physiology, Adugodi, Hosur Road, 560 030 Bangalore, India